Содержание
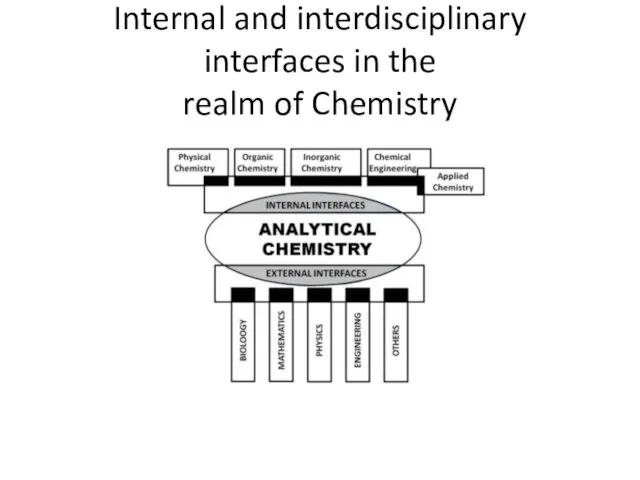
- 2. Internal and interdisciplinary interfaces in the realm of Chemistry
- 3. Analytical Chemistry Internal interfaces with other chemical areas (e.g. organic, inorganic, physical and applied chemistry, chemical
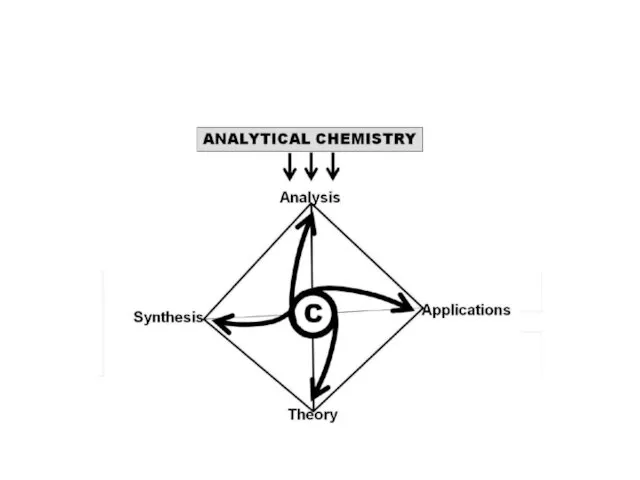
- 4. Analytical Chemistry the discipline in charge of “Analysis” (the fourth component of Chemistry in addition to
- 6. Definitions: “Analytical Chemistry is a scientific discipline that develops and applies methods, instruments and strategies to
- 7. Analytical Chemistry has two essential aims. the obtainment of as high metrological quality as possible (i.e.
- 8. Quality indicators Top or capital analytical properties Accuracy Reliability Representativeness Basic analytical properties Precision Sensitivity Selectivity
- 9. Classification of techniques by the type of analytical technique – classical or instrumental techniques; by the
- 10. Basic Equipment and Instrumentation Photo of a typical electronic balance. beaker graduated cylinder
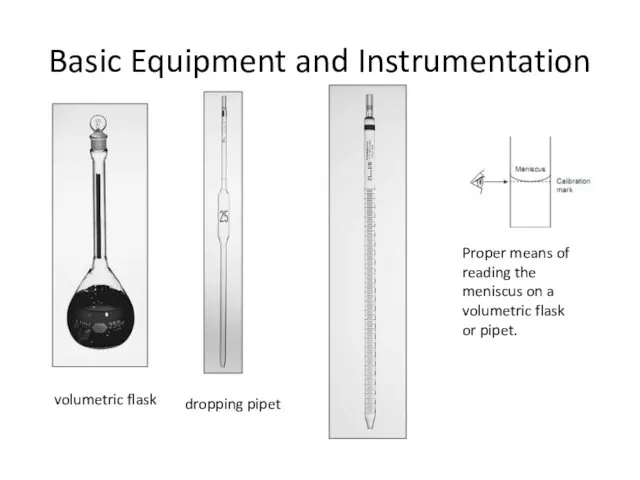
- 11. Basic Equipment and Instrumentation volumetric flask dropping pipet Proper means of reading the meniscus on a
- 12. Classical Methods: Separation of analytes by precipitation, extraction, or distillation. Qualitative analysis by reaction of analytes
- 13. Titrimetric Analysis The term titrimetric analysis refers to quantitative chemical analysis carried out by determining the
- 14. For use in titrimetric analysis a reaction must have the following conditions 1- There must be
- 15. Definition of some terms Titration is the process in which the standard reagent is added to
- 16. Indicators Indicators are often added to analyte solution in order to give an observable physical change
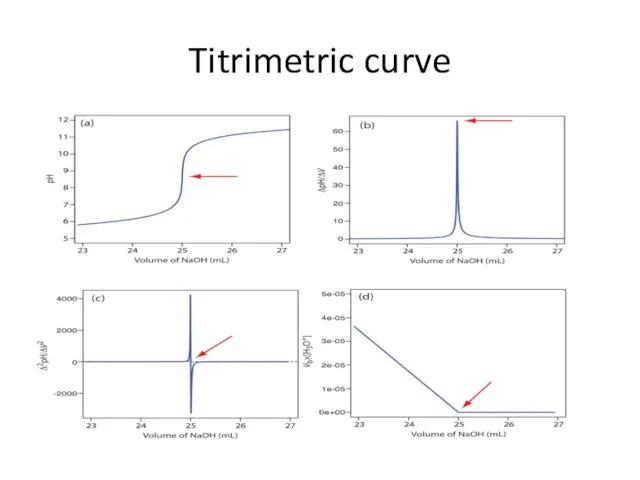
- 17. Titrimetric curve
- 18. Classification of reaction in titrimetric analysis Neutralization reaction, or acidimetry and alkalimetry. These include thetitration of
- 19. Instrumental Methods: Measurements of physical properties of analytes, such as conductivity, electrode potential, light absorption, or
- 20. The advantages of instrumental methods over classical methods include: 1. The ability to perform trace analysis,
- 21. Instrumental analysis can be further classified according to the principles by which the measurement signal is
- 22. Lists the names of instrumental methods that are based upon various analytical signals.
- 23. Electrochemical methods of analysis in which the analyte participates in a redox reaction or other process.
- 24. Spectrochemical methods of analysis in which the analyte interacts with electromagnetic radiation. Most of the methods
- 25. The technique of mass spectroscopy in which the analyte is ionized and subsequently detected. Although in
- 26. Single-Channel vs Multi-Channel Techniques single-channel techniques will generate but a single number for each analysis of
- 27. Multi-channel methods have two important advantages over their single-channel counterparts: 1. They provide the ability to
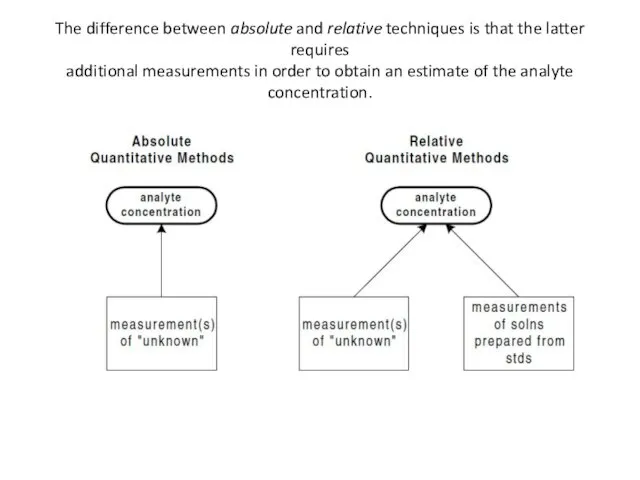
- 28. Relative vs Absolute Techniques Another way of classifying analytical techniques is according to the method by
- 29. The difference between absolute and relative techniques is that the latter requires additional measurements in order
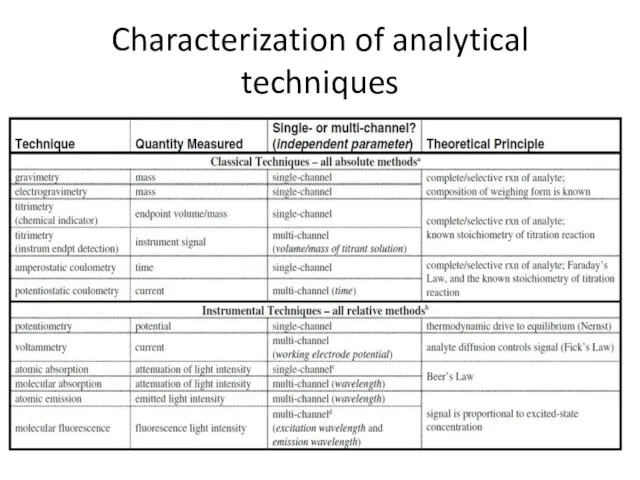
- 30. Characterization of analytical techniques
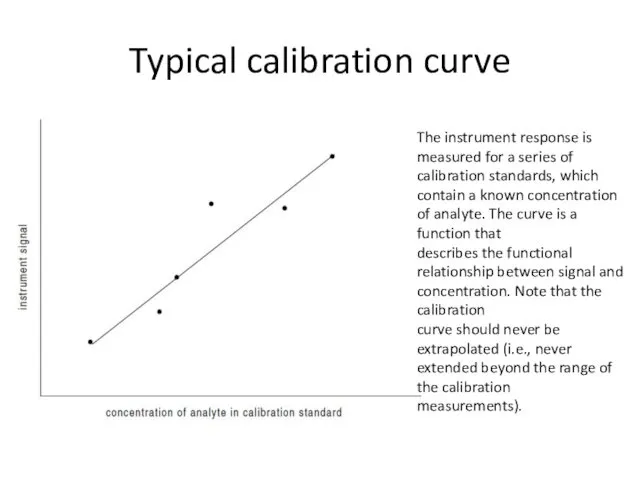
- 31. Calibration Curve Method For any instrumental method used for quantitative chemical analysis, there is some functional
- 32. Typical calibration curve The instrument response is measured for a series of calibration standards, which contain
- 33. The following points should be made about this method of quantitation: The central philosophy of the
- 34. Electroanalytical methods Electroanalytical methods are a class of techniques in analytical chemistry which study an analyte
- 36. Скачать презентацию


























 Мале Полісся
Мале Полісся Оценка радиационного фона в зоне влияния отвалов угледобывающей промышленности в Луганской области
Оценка радиационного фона в зоне влияния отвалов угледобывающей промышленности в Луганской области Особо охраняемые территории России. Заповедники
Особо охраняемые территории России. Заповедники Изменение климата Земли
Изменение климата Земли Water pollutions
Water pollutions презентация био 0804
презентация био 0804 Экологическая экспертиза
Экологическая экспертиза Методы и организация комплексного геоэкологического мониторинга
Методы и организация комплексного геоэкологического мониторинга Збережи природу
Збережи природу Экологический кризис
Экологический кризис Влияние экологических факторов Земли на растения
Влияние экологических факторов Земли на растения Экологический подход, как методология современного образования
Экологический подход, как методология современного образования Учение о биосфере презентация по экологии по учебнику Зверев А.Т. Зверева Е.Г
Учение о биосфере презентация по экологии по учебнику Зверев А.Т. Зверева Е.Г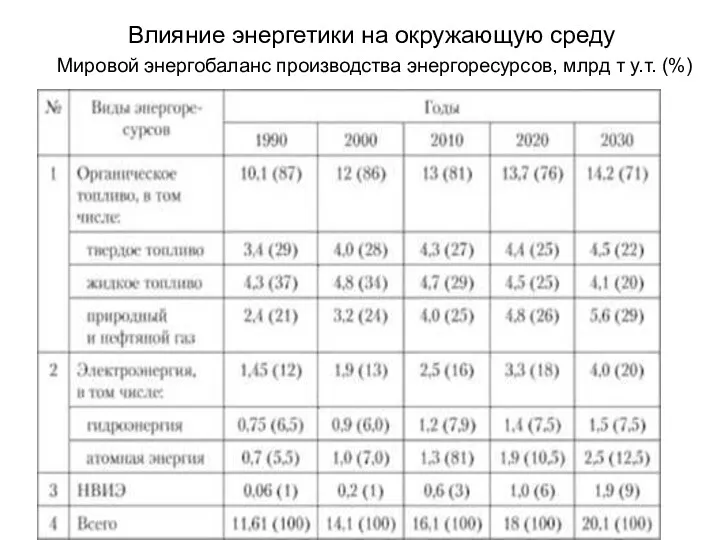 Влияние энергетики на окружающую среду. Мировой энергобаланс производства энергоресурсов
Влияние энергетики на окружающую среду. Мировой энергобаланс производства энергоресурсов Экологические проблемы городов народных республик Донбасса
Экологические проблемы городов народных республик Донбасса The pollution of lake Baikal
The pollution of lake Baikal Влияние экологии на здоровье человека
Влияние экологии на здоровье человека Санитарлық қорғау зоналары және шығарындыларды шектеу
Санитарлық қорғау зоналары және шығарындыларды шектеу Проблемы экологии в современном мире
Проблемы экологии в современном мире Правила поведения в лесу
Правила поведения в лесу Глобальные экологические проблемы современности
Глобальные экологические проблемы современности Экология Черного моря
Экология Черного моря презентация Породы деревьев произрастающие на Шаимской территории
презентация Породы деревьев произрастающие на Шаимской территории Збалансоване природокористування
Збалансоване природокористування Биосфера и человек
Биосфера и человек Основы природопользования. (Экологическое образование и воспитание.)
Основы природопользования. (Экологическое образование и воспитание.) Экологический отряд Клевер
Экологический отряд Клевер Особливості сучасної цивілізації
Особливості сучасної цивілізації