Содержание
- 2. Contents 1. Physical background of NMR 2. Chemical shielding 3. Chemical exchange 4. Dipolar interaction 5.
- 3. Books Harald Günther NMR Spectroscopy: Basic Principles, Concepts, and Applications in Chemistry James Keeler Understanding NMR
- 4. Discovery of spin and magnetic resonance effect
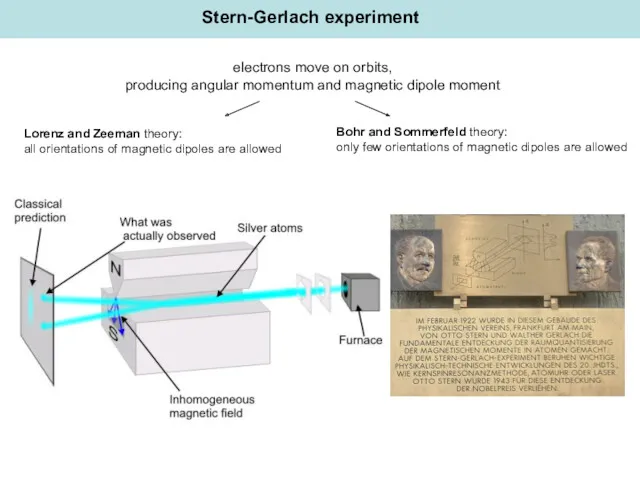
- 5. Stern-Gerlach experiment Lorenz and Zeeman theory: all orientations of magnetic dipoles are allowed Bohr and Sommerfeld
- 6. “Attached the continuation of our work (Zeitschrift für Physik 8 (1921) 110): The experimental proof of
- 7. Gerlach, W. and O. Stern, "Der experimentelle Nachweis der Richtungsquantelung". Zeitschrift fur Physik 1922, 9, 349-352.
- 8. Concept of spin (first proposed in 1925) Ralph Kronig 1904-1995 Samuel Abraham Goudsmit 1902-1978 George Uhlenbeck
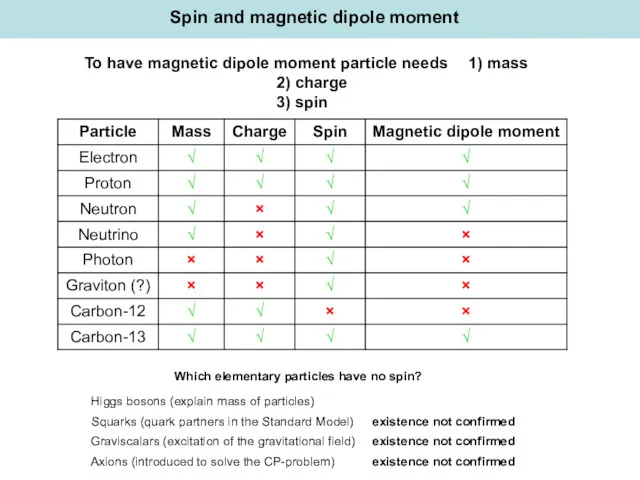
- 9. Spin and magnetic dipole moment To have magnetic dipole moment particle needs 1) mass 2) charge
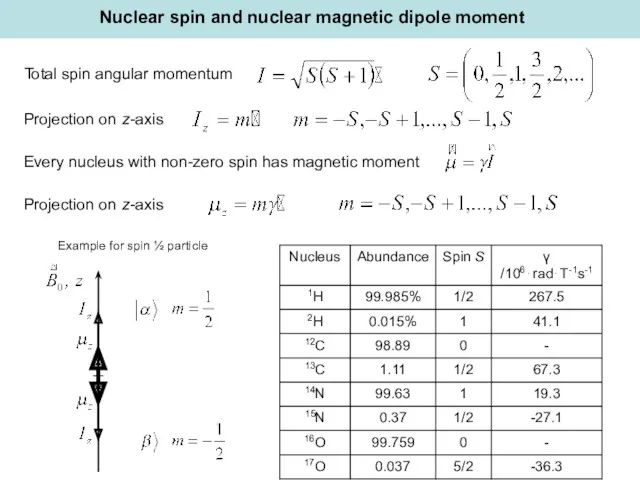
- 10. Total spin angular momentum Projection on z-axis Nuclear spin and nuclear magnetic dipole moment Every nucleus
- 11. Nuclear spin 1/2 in magnetic field
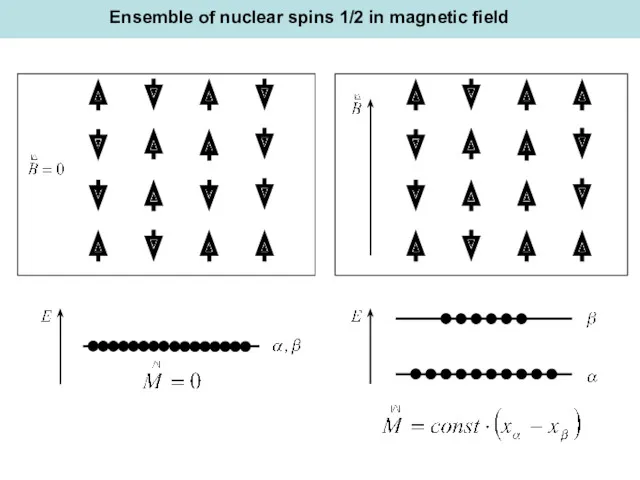
- 12. Ensemble of nuclear spins 1/2 in magnetic field
- 13. Isidor Isaac Rabi 1898-1988 Columbia U., NY, USA 1944 Felix Bloch 1905-1983 Stanford U., CA, USA
- 14. Nobel Prizes for the applications of NMR Richard R. Ernst b. 1933 ETH, Switzerland 1991 Multidimensional
- 15. What are the reasons behind NMR's success?... nature has generously provided us with three basic physical
- 16. Overview of an NMR experiment and design of NMR instruments
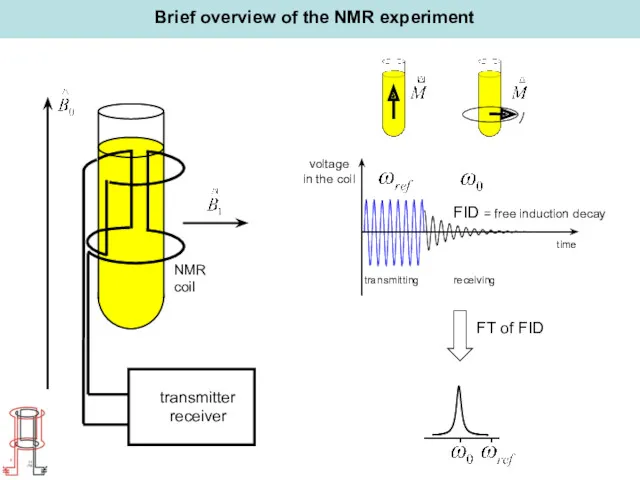
- 17. Brief overview of the NMR experiment transmitting receiving voltage in the coil time FID = free
- 19. Скачать презентацию











 Звуковые волны
Звуковые волны Химия окружающей среды. Радиоактивные элементы в окружающей среде
Химия окружающей среды. Радиоактивные элементы в окружающей среде Магнитное поле в веществе
Магнитное поле в веществе Перемещение при прямолинейном равномерном движении
Перемещение при прямолинейном равномерном движении Простой механизм. Двигатель
Простой механизм. Двигатель Законы сохранения в механике
Законы сохранения в механике Монтаж гидроагрегата. Монтаж гидротурбины
Монтаж гидроагрегата. Монтаж гидротурбины Өлшеу қателігі. Құрал шкаласының және жүйелік қателерді есепке алу. Қос қателікті бағалау
Өлшеу қателігі. Құрал шкаласының және жүйелік қателерді есепке алу. Қос қателікті бағалау презентация 10 класс Космическая скорость
презентация 10 класс Космическая скорость Силы в природе
Силы в природе Шекаралық қабат теңдеулерінің жуық шешімдері
Шекаралық қабат теңдеулерінің жуық шешімдері Домашнее задание по курсу Детали машин №4: Проверочный расчет тихоходного вала редуктора
Домашнее задание по курсу Детали машин №4: Проверочный расчет тихоходного вала редуктора Постулаты Н. Бора
Постулаты Н. Бора Многосигнальная селективность магистральных радиоприемных устройств в декаметровом диапазоне волн
Многосигнальная селективность магистральных радиоприемных устройств в декаметровом диапазоне волн Распределение Больцмана. (Лекция 10)
Распределение Больцмана. (Лекция 10) Детали машин и подъемное оборудование. Цепные передачи
Детали машин и подъемное оборудование. Цепные передачи Ядерная физика
Ядерная физика Расчет и анализ электромагнитных полей элементарных излучателей
Расчет и анализ электромагнитных полей элементарных излучателей Инновационный проект производства USB-розеток
Инновационный проект производства USB-розеток Основы физики деления ядер
Основы физики деления ядер Загальна будова додаткового обладнання вантажного автомобіля
Загальна будова додаткового обладнання вантажного автомобіля Полупроводники
Полупроводники Радиационные испытания материалов и изделий
Радиационные испытания материалов и изделий Радиационные термометры
Радиационные термометры Разработка технологического процесса механической обработки и проектирование участка механического цеха детали Фланец 03
Разработка технологического процесса механической обработки и проектирование участка механического цеха детали Фланец 03 Электромагнитные волны и их свойства. Шкала электромагнитных волн
Электромагнитные волны и их свойства. Шкала электромагнитных волн Коробки передач. Требования
Коробки передач. Требования Робототехника. Робот-дворник
Робототехника. Робот-дворник