Содержание
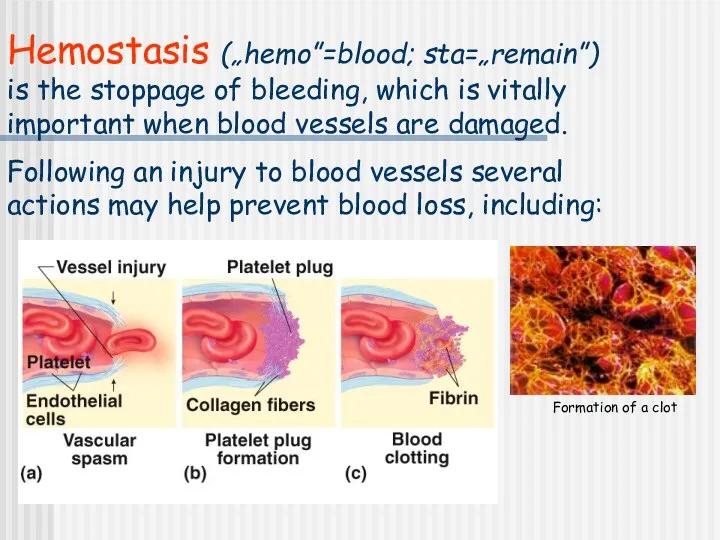
- 2. Hemostasis („hemo”=blood; sta=„remain”) is the stoppage of bleeding, which is vitally important when blood vessels are
- 3. Local vasoconstriction is due to local spasm of the smooth muscle (symp. reflex) can be maintained
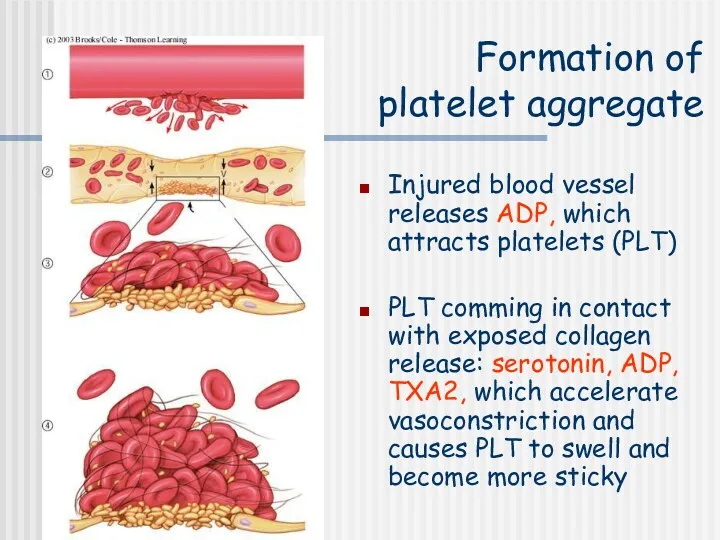
- 4. Formation of platelet aggregate Injured blood vessel releases ADP, which attracts platelets (PLT) PLT comming in
- 5. The micrograph shows activated platelets adhering to some damaged cells
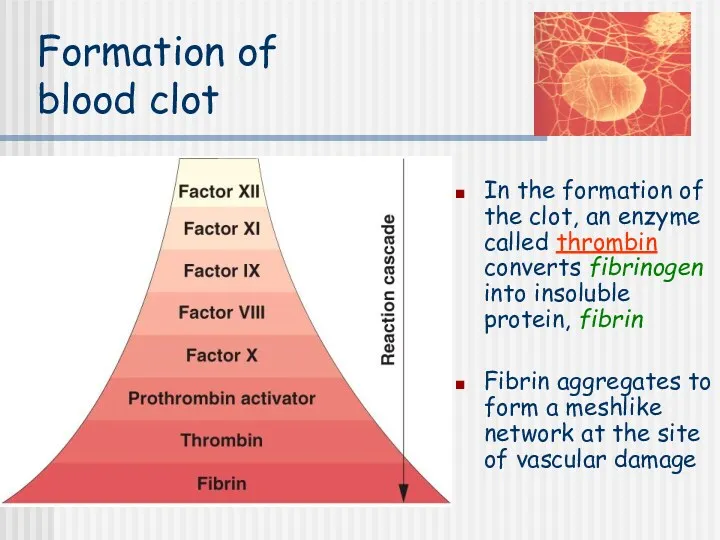
- 6. Formation of blood clot In the formation of the clot, an enzyme called thrombin converts fibrinogen
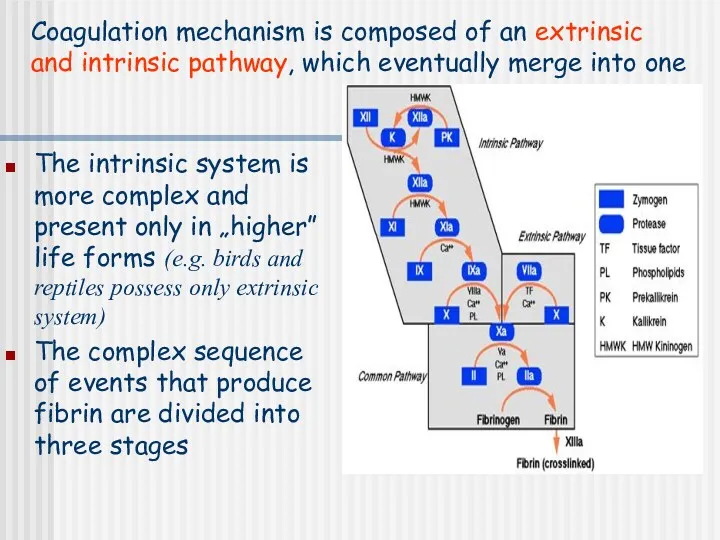
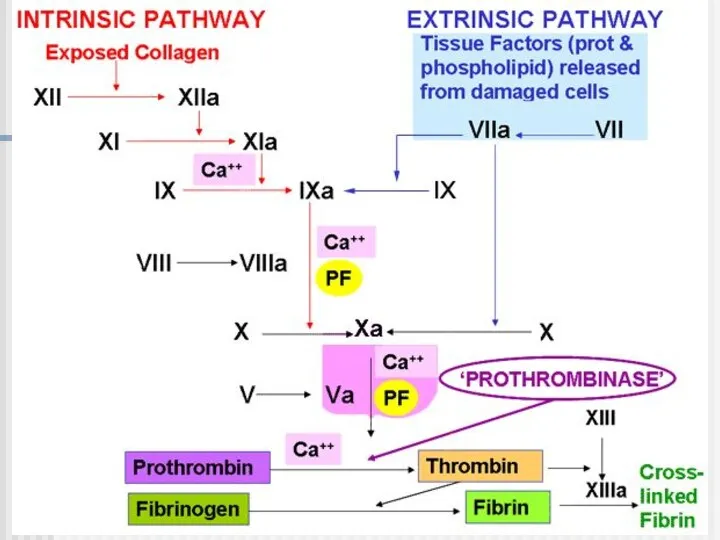
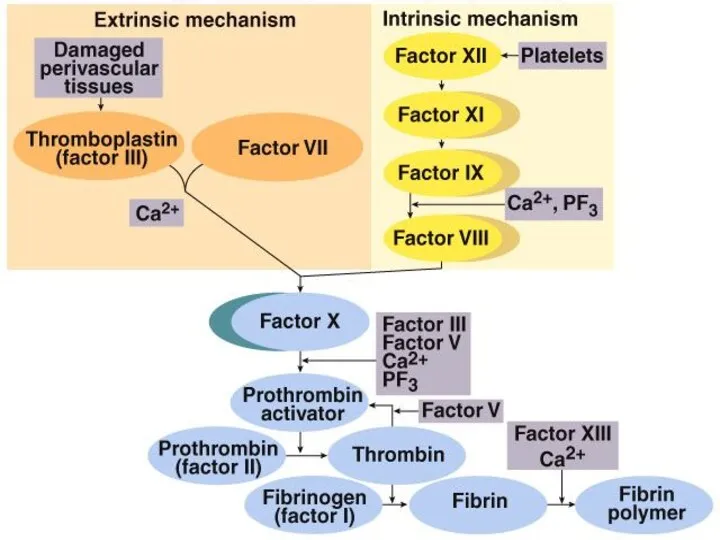
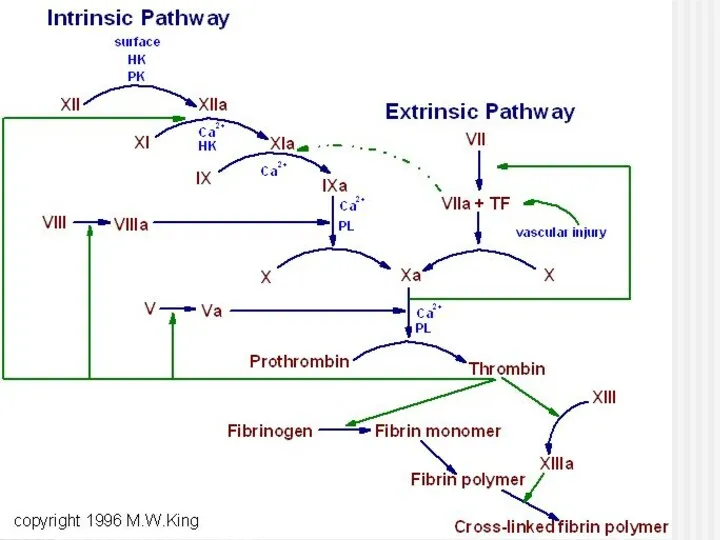
- 7. The intrinsic system is more complex and present only in „higher” life forms (e.g. birds and
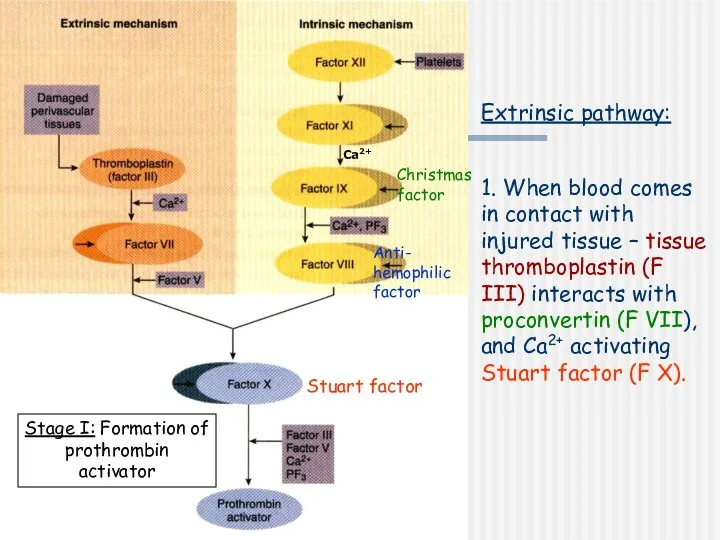
- 8. Extrinsic pathway: 1. When blood comes in contact with injured tissue – tissue thromboplastin (F III)
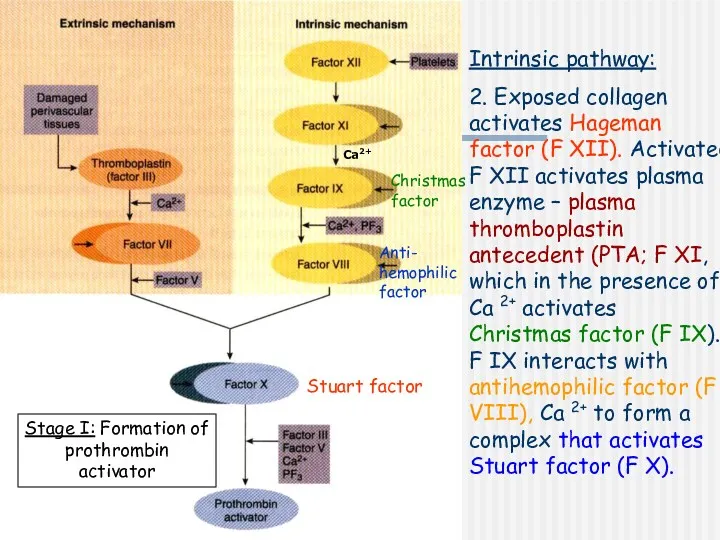
- 9. Intrinsic pathway: 2. Exposed collagen activates Hageman factor (F XII). Activated F XII activates plasma enzyme
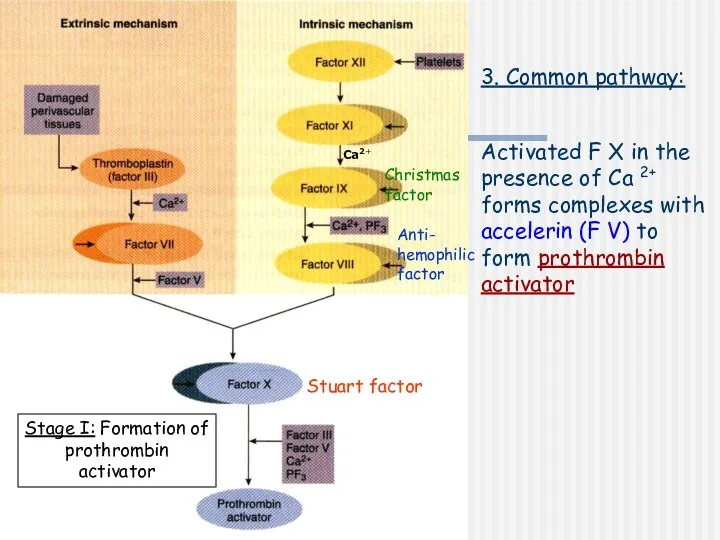
- 10. Stage I: Formation of prothrombin activator 3. Common pathway: Activated F X in the presence of
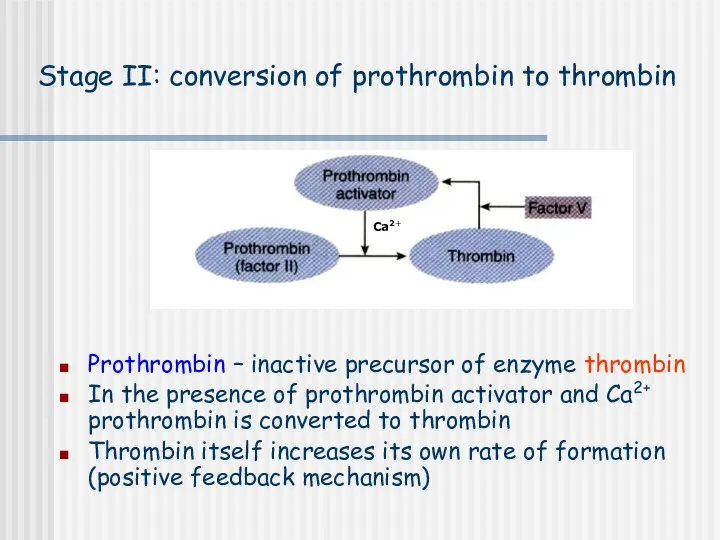
- 12. Stage II: conversion of prothrombin to thrombin Prothrombin – inactive precursor of enzyme thrombin In the
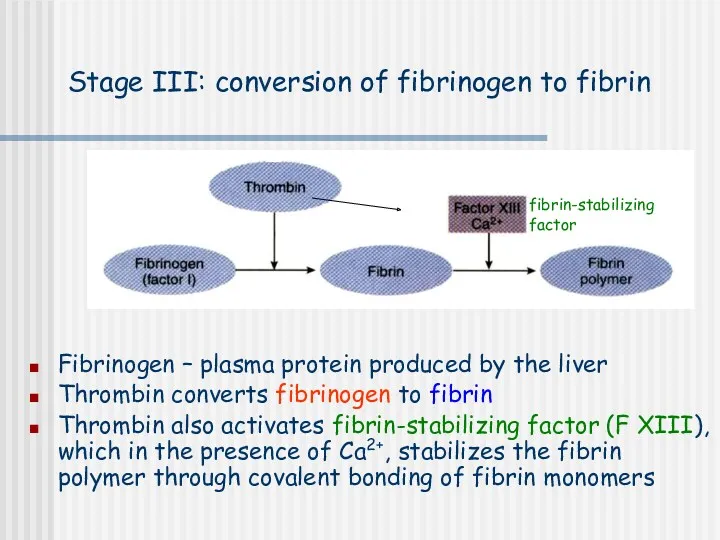
- 13. Stage III: conversion of fibrinogen to fibrin Fibrinogen – plasma protein produced by the liver Thrombin
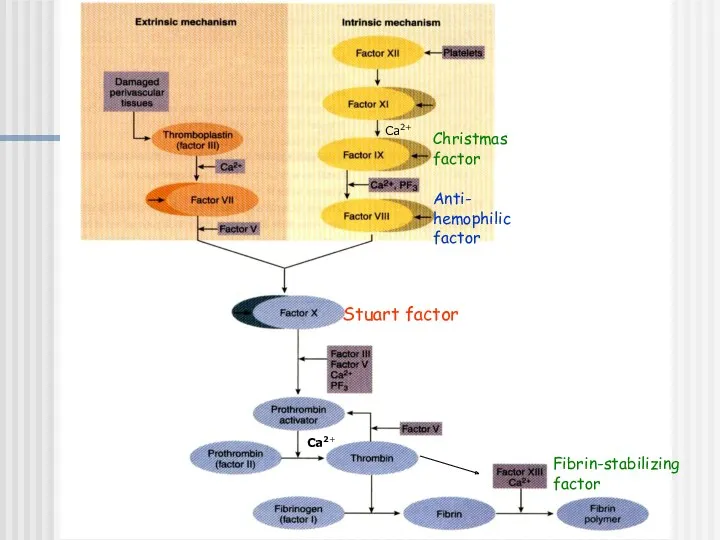
- 14. Calcium ions Are required for promotion and acceleration of almost all blood clotting reactions Except: activation
- 15. Ca2+ Ca2+ Christmas factor Anti- hemophilic factor Stuart factor Fibrin-stabilizing factor
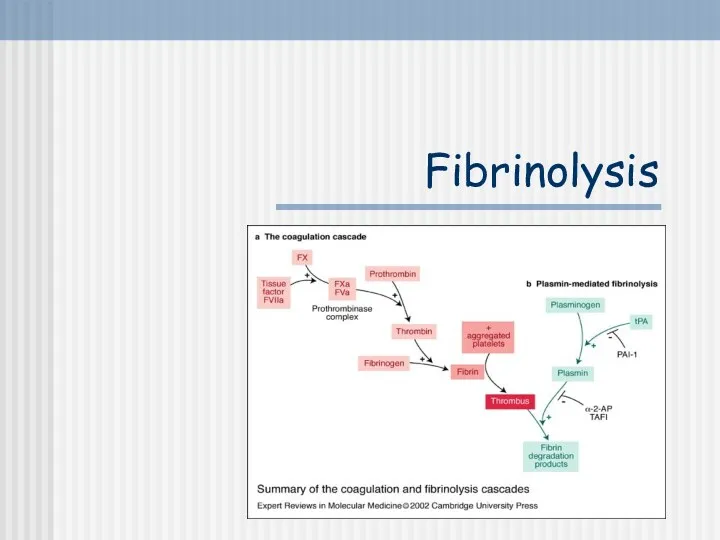
- 18. Fibrinolysis
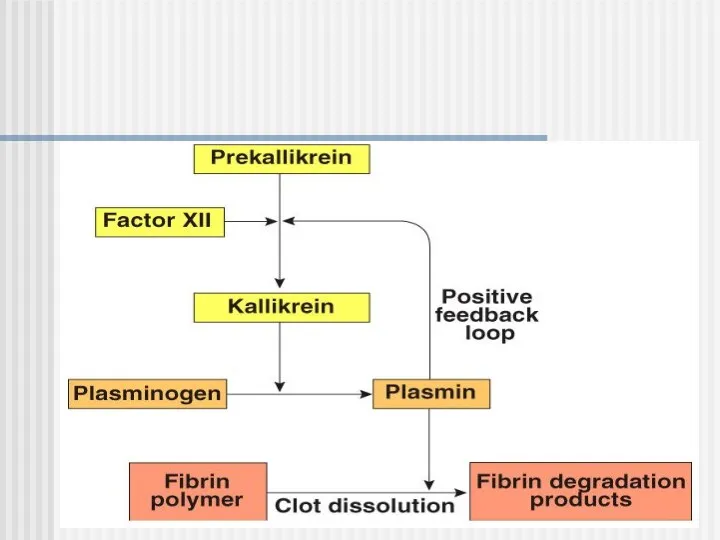
- 19. Clot Dissolution Plasmin is formed from plasminogen - enzyme called activator (e.g. enzymes from urine, tears,
- 21. Anticoagulants Hirudo medicinalis produce Hirudin that inhibits Thrombin
- 22. Anticoagulants Although tissue breakdown and platelets destruction are normal events in the absence of trauma, intravascular
- 23. Natural anticoagulants Antithrombin III – inhibits factor X and thrombin Heparin from basophils and mast cells
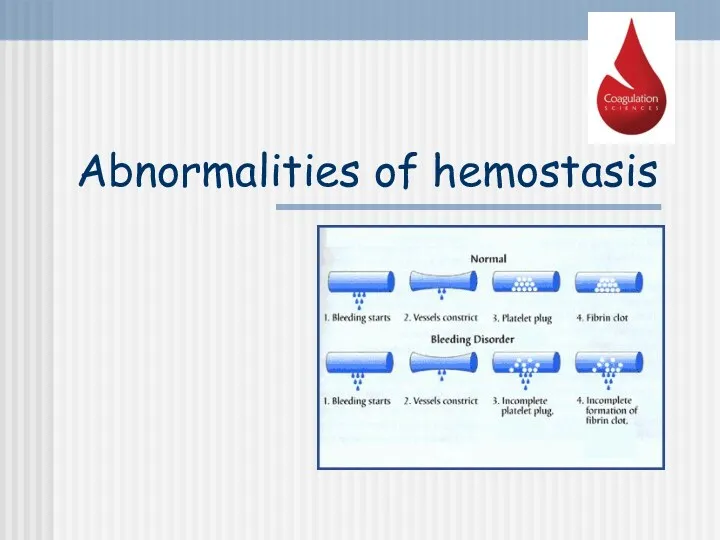
- 24. Abnormalities of hemostasis
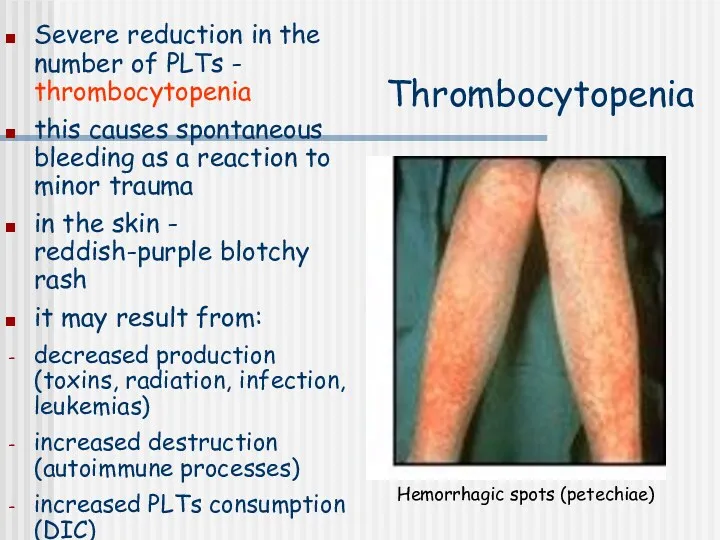
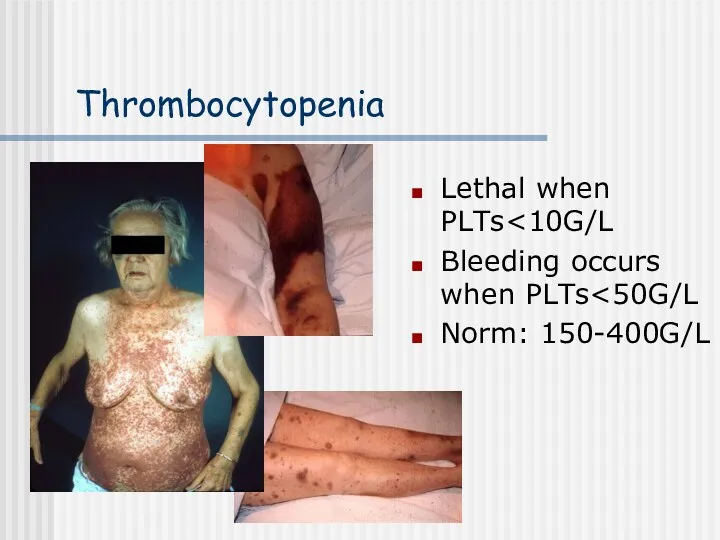
- 25. Thrombocytopenia Severe reduction in the number of PLTs - thrombocytopenia this causes spontaneous bleeding as a
- 26. Thrombocytopenia Lethal when PLTs Bleeding occurs when PLTs Norm: 150-400G/L
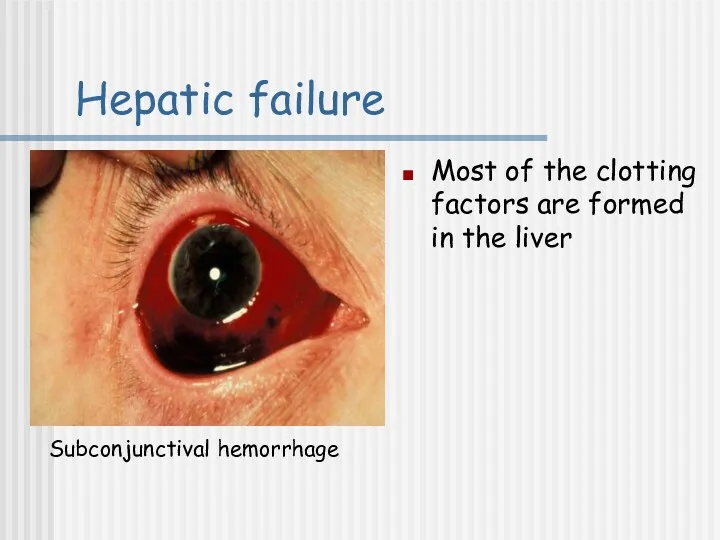
- 27. Hepatic failure Most of the clotting factors are formed in the liver Subconjunctival hemorrhage
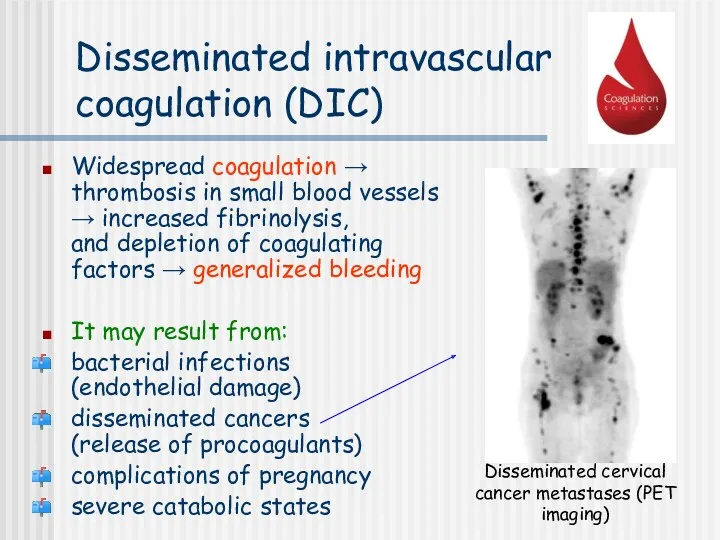
- 28. Disseminated intravascular coagulation (DIC) Widespread coagulation → thrombosis in small blood vessels → increased fibrinolysis, and
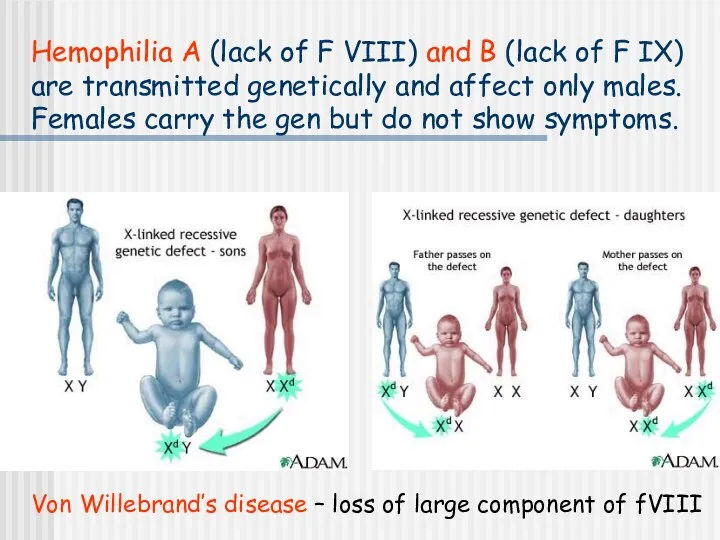
- 29. Hemophilia A (lack of F VIII) and B (lack of F IX) are transmitted genetically and
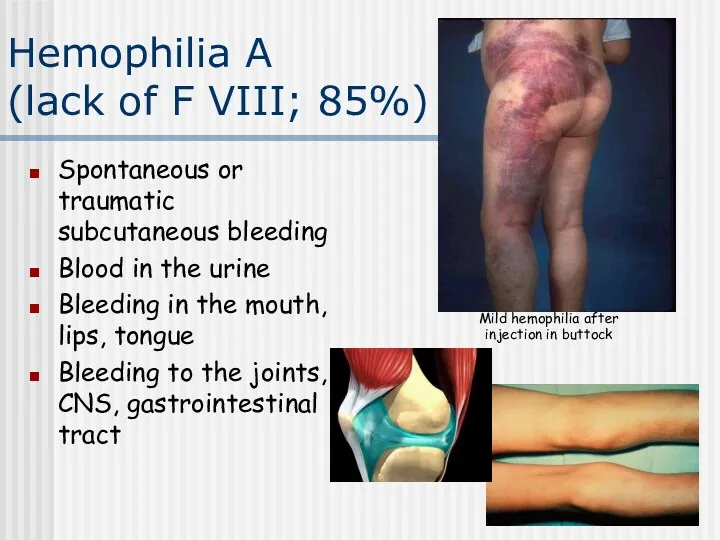
- 30. Hemophilia A (lack of F VIII; 85%) Spontaneous or traumatic subcutaneous bleeding Blood in the urine
- 31. Son of the last Tsar of Russia – Aleksy Romanow suffered from Hemophilia A
- 32. Tests of coagulation
- 33. "Intrinsic" and "extrinsic" coagulation pathways N: 9.9 – 13 sec Activated Partial Thromboplastin Time N: 25-35
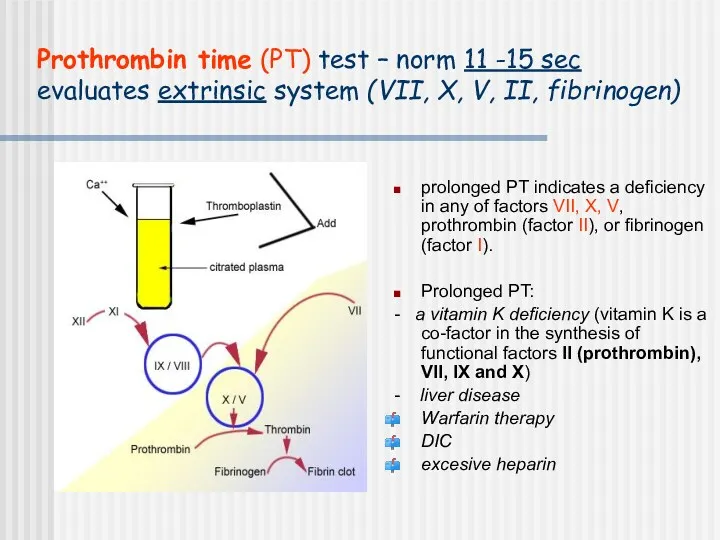
- 34. Prothrombin time (PT) test – norm 11 -15 sec evaluates extrinsic system (VII, X, V, II,
- 35. International Normalised Ratio (INR) The result for the PT is expressed as a ratio (prothrombin clotting
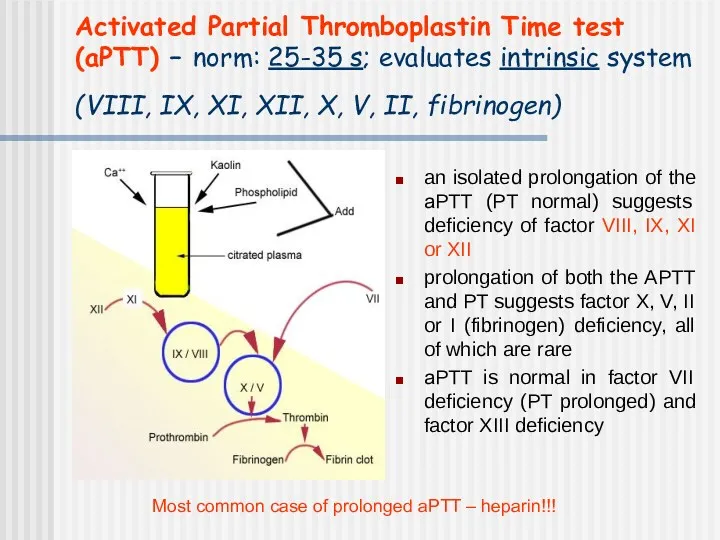
- 36. Activated Partial Thromboplastin Time test (aPTT) – norm: 25-35 s; evaluates intrinsic system (VIII, IX, XI,
- 37. Thrombin time (TT) – norm: 14-15 sec Prolonged TT: Heparin (much more sensitive to heparin than
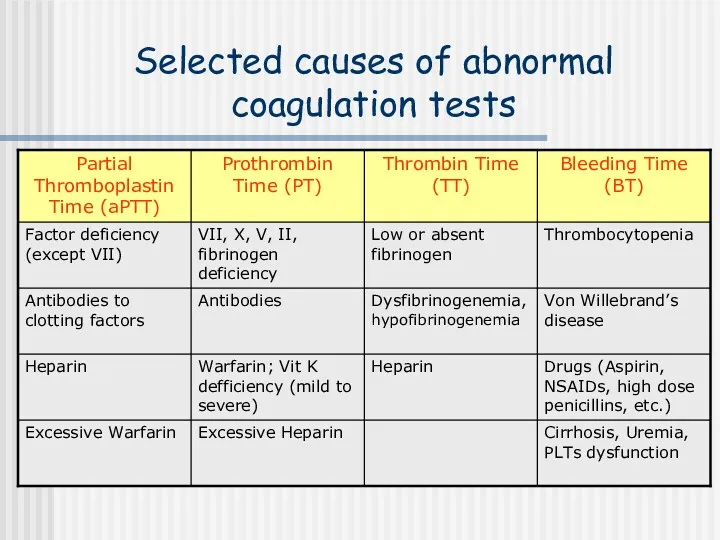
- 38. Selected causes of abnormal coagulation tests
- 39. "Intrinsic" and "extrinsic" coagulation pathways N: 9.9 – 13 sec Activated Partial Thromboplastin Time N: 25-35
- 40. Whole blood clotting time The time taken for blood to clot mainly reflects the time required
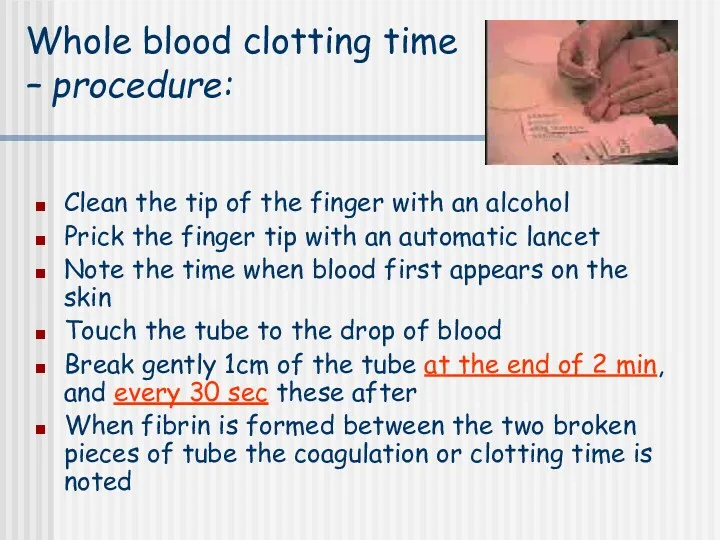
- 41. Whole blood clotting time – procedure: Clean the tip of the finger with an alcohol Prick
- 42. Bleeding time This is a test that measures the speed in which small blood vessels close
- 43. Bleeding time – procedure: Clean the earlobe with an alcohol Prick the earlobe with an automatic
- 44. Abnormal Bleeding Time Prolonged bleeding time may indicate: A vascular (blood vessel) defect A platelet function
- 45. http://www.medicine.mcgill.ca/physio/vlab212D/bloodlab/images/clottime5.mpg
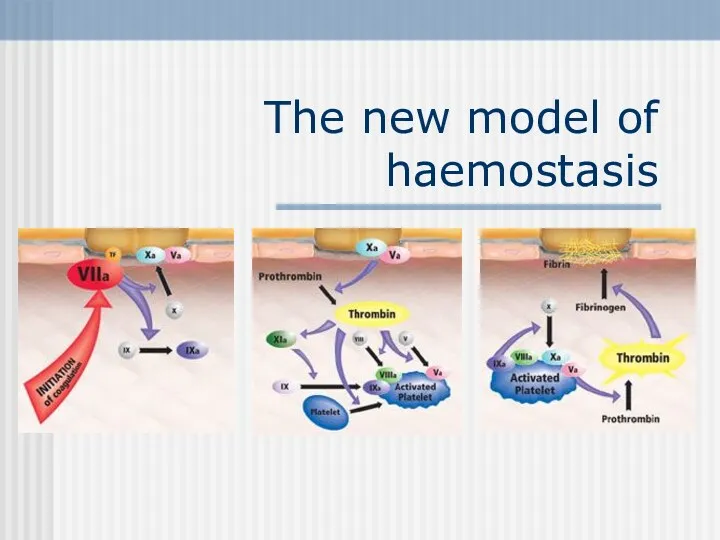
- 47. The new model of haemostasis
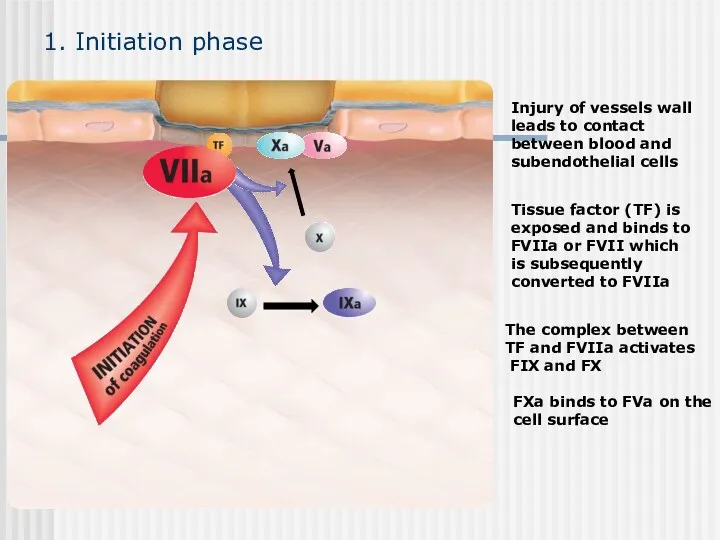
- 48. Injury of vessels wall leads to contact between blood and subendothelial cells FXa binds to FVa
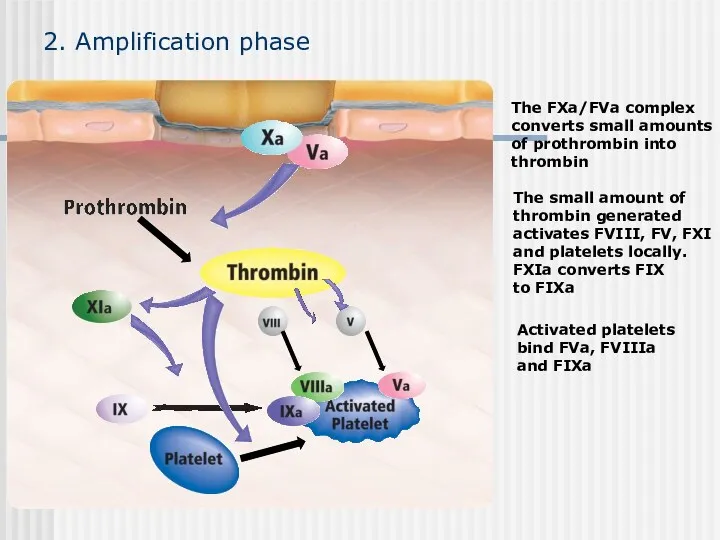
- 49. 2. Amplification phase Activated platelets bind FVa, FVIIIa and FIXa
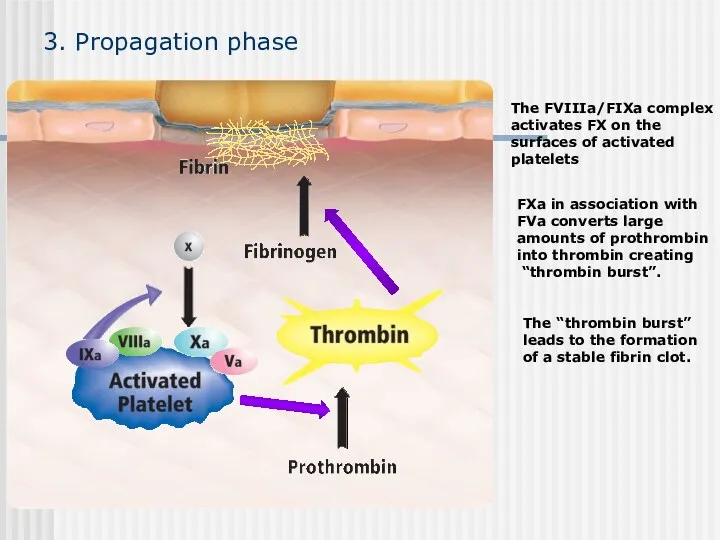
- 50. 3. Propagation phase The “thrombin burst” leads to the formation of a stable fibrin clot.
- 51. Summary: Haemostasis starts with the interaction between TF and FVIIa on the surface of subendothelial cells.
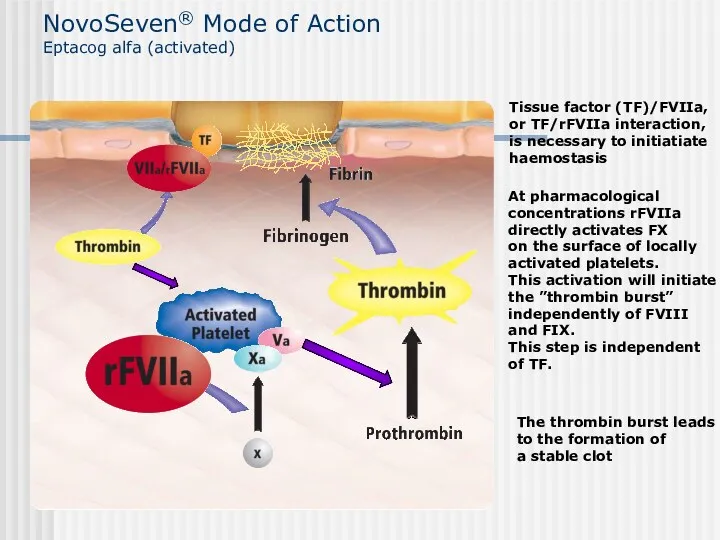
- 52. NovoSeven® Mode of Action Eptacog alfa (activated) The thrombin burst leads to the formation of a
- 53. Conclusion: • In high doses rFVIIa binds to the surface of the locally activated platelets where
- 54. Prescribing Information NovoSeven® Eptacog alfa (activated) Abbreviated Prescribing Information: NovoSeven [Recombinant Coagulation Factor VIIa (rFVIIa)] Presentation:
- 55. A 35-year-old man complains of chronic physical fatigue, which began 3-4 weeks ago. He said he
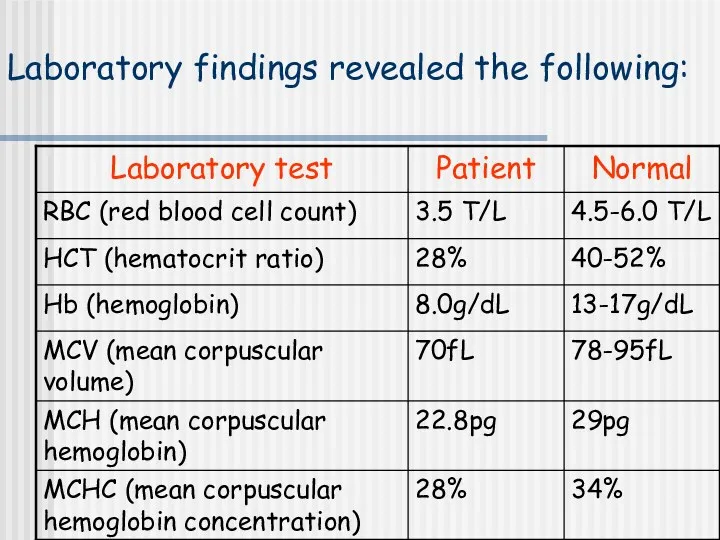
- 56. Laboratory findings revealed the following:
- 57. Case history questions: What general medical condition is suggested by the person’s symptoms? What fundamental change
- 59. Скачать презентацию
























 Плевральный выпот
Плевральный выпот Заболевания органов дыхания у детей
Заболевания органов дыхания у детей Пневмония. Этиология. Принципы лечения
Пневмония. Этиология. Принципы лечения Тактика наружной терапии атопического дерматита: современный взгляд
Тактика наружной терапии атопического дерматита: современный взгляд Мерез. Мерез ауруының өзектілігі
Мерез. Мерез ауруының өзектілігі Дәлелді медицина – дәрілік заттарды қолдану құралы түрінде. Дәрілік заттарды қате жарнамалаудың белгілері
Дәлелді медицина – дәрілік заттарды қолдану құралы түрінде. Дәрілік заттарды қате жарнамалаудың белгілері Диагностика и малоинвазивные методы лечения жидкостных образований брюшной полости
Диагностика и малоинвазивные методы лечения жидкостных образований брюшной полости Ортодонтическое лечение
Ортодонтическое лечение Современные аспекты комплексного сестринского ухода за пациентами с хроническим гастритом
Современные аспекты комплексного сестринского ухода за пациентами с хроническим гастритом Наркоз (общее обезболивание, общая анестезия)
Наркоз (общее обезболивание, общая анестезия) Современные алгоритмы лечения сахарного диабета 2 типа
Современные алгоритмы лечения сахарного диабета 2 типа Иммунопатология и аллергия
Иммунопатология и аллергия Лучевая диагностика заболеваний пищеварительного тракта
Лучевая диагностика заболеваний пищеварительного тракта Анафилактический шок
Анафилактический шок Психолого-медико-педагогическая комиссия
Психолого-медико-педагогическая комиссия Выделительная система
Выделительная система Заболевания органов мочевыводящей системы
Заболевания органов мочевыводящей системы Хронический холецистит. Классификация. Диагностика и лечение некалькулезного и калькулезного холецистита
Хронический холецистит. Классификация. Диагностика и лечение некалькулезного и калькулезного холецистита Омыртқаның зақымдалуы.Жіктемесі. диагностикасы.емі
Омыртқаның зақымдалуы.Жіктемесі. диагностикасы.емі Кора полушарий головного мозга. Расстройства высших корковых функций
Кора полушарий головного мозга. Расстройства высших корковых функций Об утверждении санитарных норм СанПин противоэпидемических мероприятий в том числе и работе с медицинскими отходами
Об утверждении санитарных норм СанПин противоэпидемических мероприятий в том числе и работе с медицинскими отходами Предмет и задачи токсикологии
Предмет и задачи токсикологии Доброкачественные и злокачественные новообразования вульвы
Доброкачественные и злокачественные новообразования вульвы Железодефицитные анемии у новорожденных современные возможности профилактики и лечения
Железодефицитные анемии у новорожденных современные возможности профилактики и лечения Геморрагиялық шок
Геморрагиялық шок Спадкові хвороби
Спадкові хвороби Организация медицинского обеспечения населения в чрезвычайных ситуациях
Организация медицинского обеспечения населения в чрезвычайных ситуациях СӨЖ. Глаукома
СӨЖ. Глаукома