Содержание
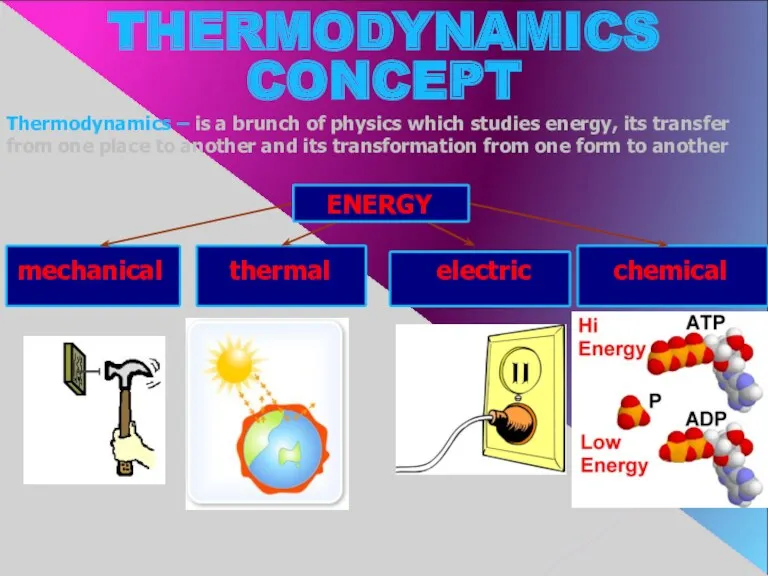
- 2. Thermodynamics – is a brunch of physics which studies energy, its transfer from one place to
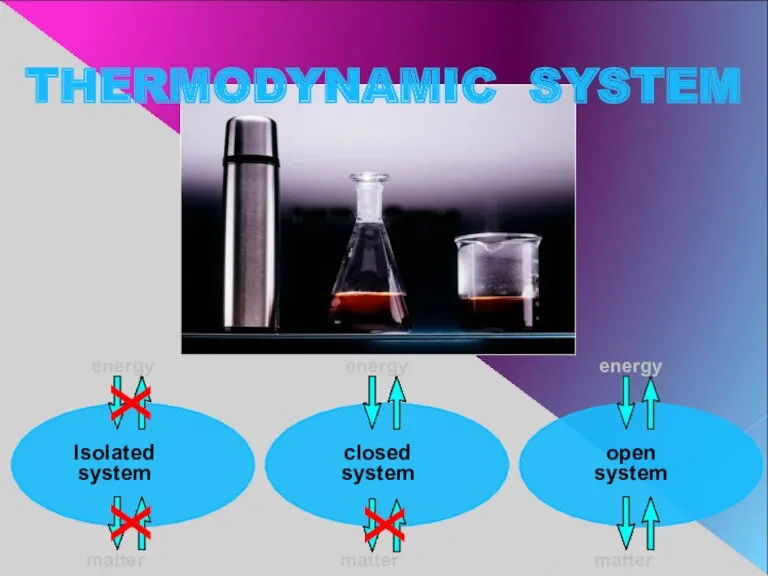
- 3. Isolated system closed system open system energy energy energy matter matter matter THERMODYNAMIC SYSTEM х х
- 4. ISOLATED SYSTEM This system doesn’t exchange energy or matter with the surroundings. Universe is considered to
- 5. CLOSED SYSTEM A system, that doesn’t exchange matter but exchanges energy with the surroundings.
- 6. OPENED SYSTEM A system, that exchanges both matter and energy with surroundings. All living organisms are
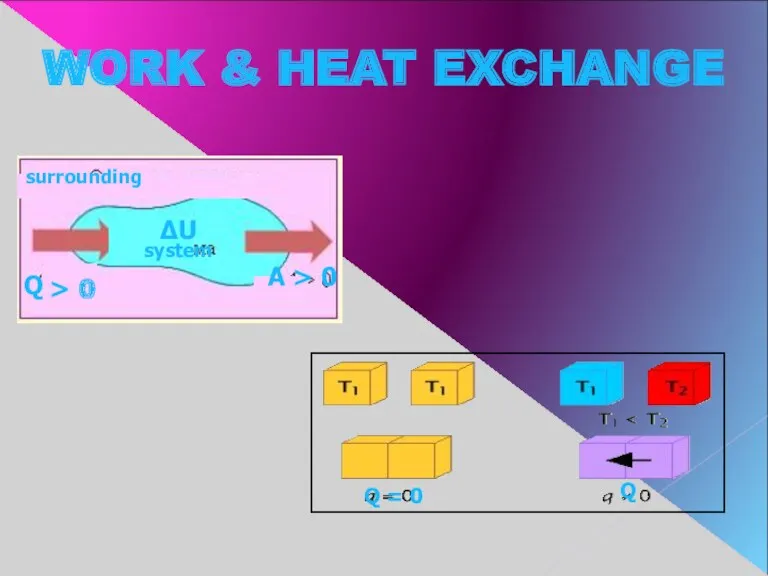
- 7. WORK & HEAT EXCHANGE > 0 surrounding Q
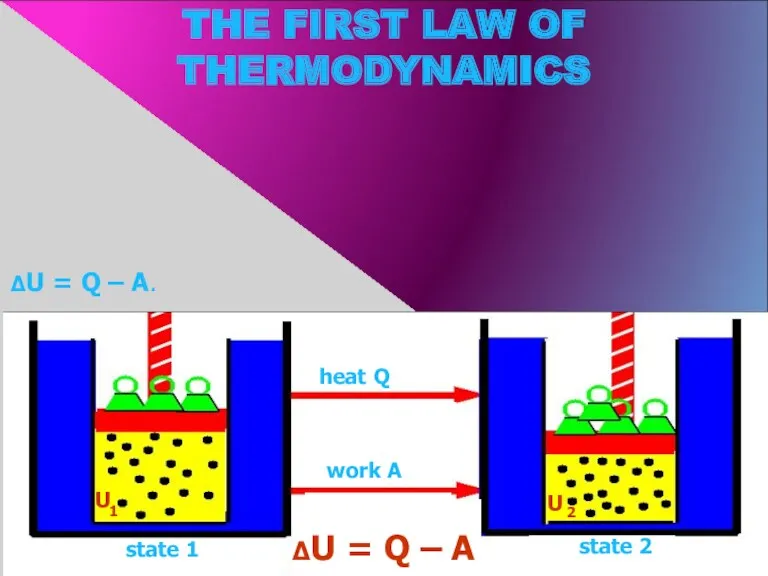
- 8. THE FIRST LAW OF THERMODYNAMICS state 1 state 2 heat Q work А U U 1
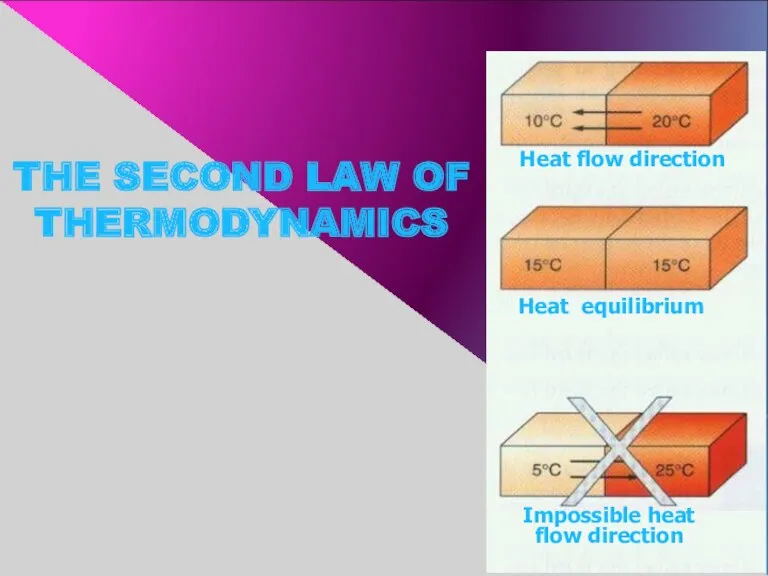
- 9. THE SECOND LAW OF THERMODYNAMICS Heat equilibrium Heat flow direction Impossible heat flow direction
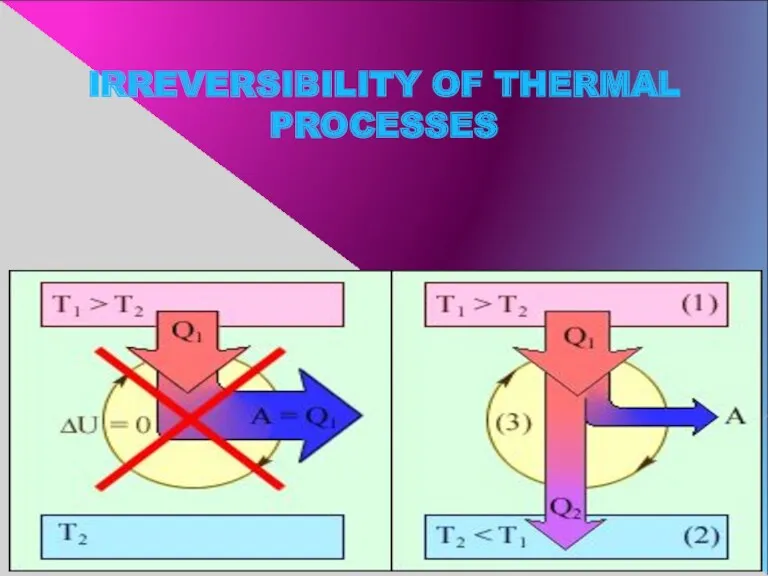
- 10. IRREVERSIBILITY OF THERMAL PROCESSES
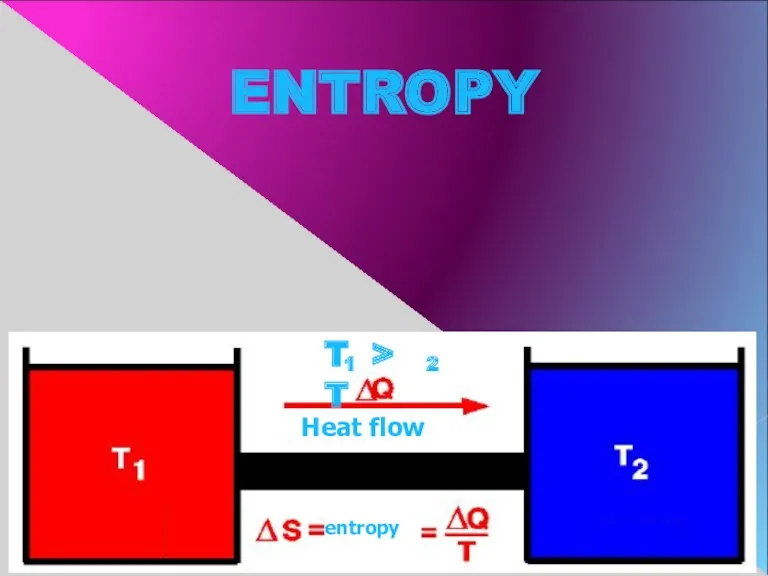
- 11. ENTROPY Heat flow entropy T > T 1 2
- 12. Prigogine state ΔiS ΔвS ΔeS Open thermodynamic system
- 13. BIOCALORYMETRY Biocalorymetry – is a measurement of the energetics of biological processes such as biochemical reactions,
- 14. DIRECT BIOCALORYMETRY Inside the Atwater-Benedict human calorimeter.
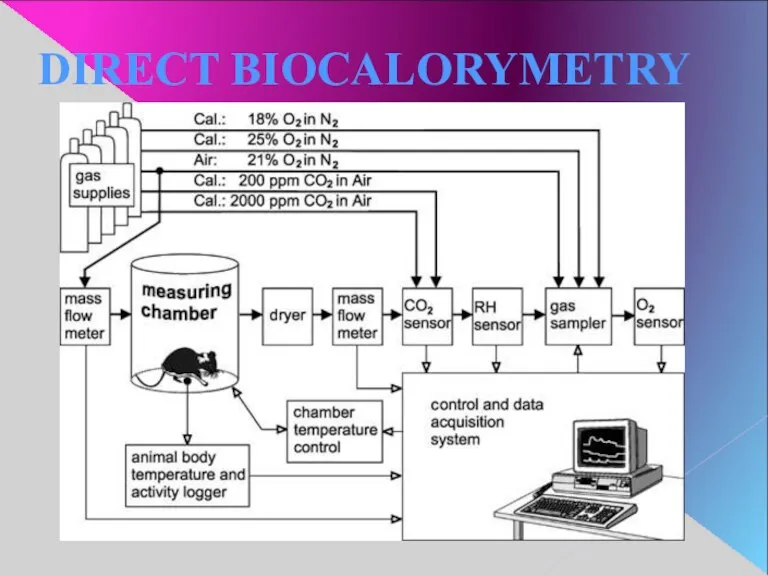
- 15. DIRECT BIOCALORYMETRY
- 16. INDIRECT BIOCALORYMETRY CALORIMETER
- 18. Скачать презентацию







 Размножение и оплодотворение у растений. Двойное оплодотворение у цветковых растений
Размножение и оплодотворение у растений. Двойное оплодотворение у цветковых растений Царство Вирусы
Царство Вирусы Презентация к уроку зоологии
Презентация к уроку зоологии Отряд Журавлеобразные
Отряд Журавлеобразные Микробиология
Микробиология Ах, Картошка объеденье
Ах, Картошка объеденье Самые страшные животные мира
Самые страшные животные мира Презентация к уроку по биологии на тему Иглокожие 7 класс
Презентация к уроку по биологии на тему Иглокожие 7 класс Клонування організмів
Клонування організмів Биология и проблема пищевых ресурсов
Биология и проблема пищевых ресурсов Класс Двустворчатые моллюски
Класс Двустворчатые моллюски Групи грибів
Групи грибів Животный мир крайнего Севера
Животный мир крайнего Севера Методы физиологических исследований
Методы физиологических исследований Кожа. Строение кожи
Кожа. Строение кожи Неврология. Нервная система
Неврология. Нервная система Презентация к занятию по теме Аллергия
Презентация к занятию по теме Аллергия Презентация урока Отдел Моховидные. Тайны маленьких растений
Презентация урока Отдел Моховидные. Тайны маленьких растений Ненаследственная изменчивость
Ненаследственная изменчивость Царство грибы
Царство грибы презентация к уроку биологии по теме Клетка
презентация к уроку биологии по теме Клетка Взаємодії організмів у екосистемах
Взаємодії організмів у екосистемах Система крови
Система крови Биология размножения и развития
Биология размножения и развития Професія дощового черв’яка
Професія дощового черв’яка Презентация по биологии для учащихся 7 класса по теме Класс Пресмыкающиеся
Презентация по биологии для учащихся 7 класса по теме Класс Пресмыкающиеся Вид. Критерии вида
Вид. Критерии вида Человек. Загадка человека
Человек. Загадка человека