Product Overview :
Is it a prescription product ? Yes /
No
Who is the prescriber ? (Gynecologist, Doctor, other specialist …)
Do the pharmacists shall respect the prescription ? Yes / No
Is it a Substitution market ? Yes / No
Is the product reimbursed ? Yes / No
Where the product could be sold ? (Pharmacies, Doctors, hospitals…)
Should you promote the products ?
If yes, to whom should you promote the product ? (Prescriber / Pharmacists …)
Do you have medical representatives ? Yes / No ; How many ?
If yes, in which sector ? (Gynecology, Oncology …)
Is European packaging accepted ? Yes / No
Should we use a dedicated packaging ? Yes / No
PRODUCT GENERAL INFO
Registration process :
Which type of stability study ICH is required ? (Zone II, IVa, IVb …)
Registration timeline ? (12 months, 24 months …)
Registration fees ?
Is the product price setting free or fixed by the MOH ?
If it’s fixed, please explain the process:



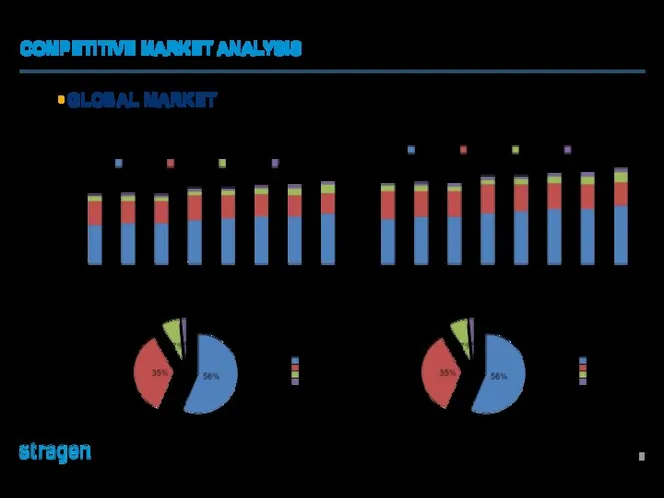
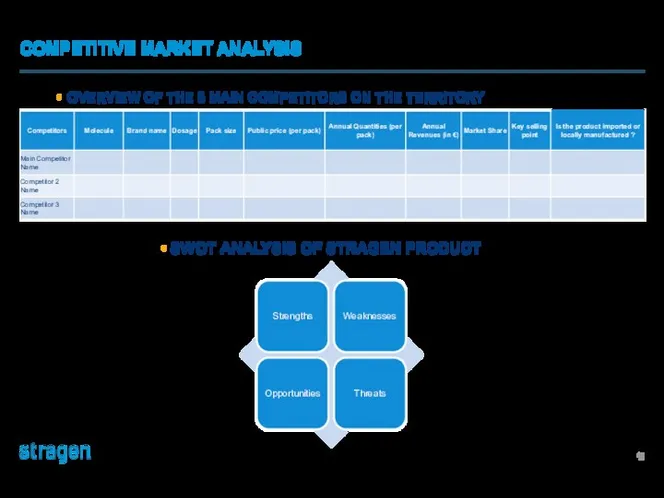
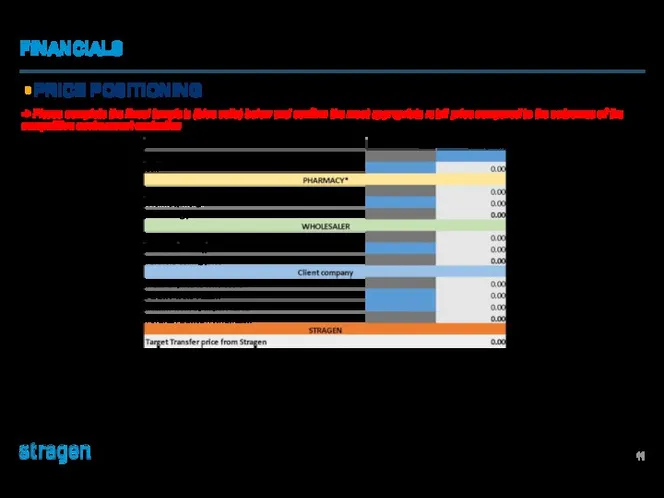

 Разработка рекламного продукта Bad Boys
Разработка рекламного продукта Bad Boys Путь к полнодозовой комбинированной терапии АГ
Путь к полнодозовой комбинированной терапии АГ Компания по производству одежды
Компания по производству одежды Прайс лист (Основной)
Прайс лист (Основной) Auchan InExtenso. Bébé Conf Boy Saison S23
Auchan InExtenso. Bébé Conf Boy Saison S23 Академия Вэлнэс. Продукция
Академия Вэлнэс. Продукция Сегментация рынка. Лекция 5
Сегментация рынка. Лекция 5 Разработка и технологии производства рекламного и PR-продукта. Рекламный лозунг
Разработка и технологии производства рекламного и PR-продукта. Рекламный лозунг Управление конкурентоспособностью в международной компании
Управление конкурентоспособностью в международной компании Alango technologies
Alango technologies Resume. Как устроен интернет
Resume. Как устроен интернет Логотип сети Теплые и пышные
Логотип сети Теплые и пышные Инновационный бизнес - продукт. Your iron toy
Инновационный бизнес - продукт. Your iron toy Creativity: some myths debunked
Creativity: some myths debunked ООО Саратовская макаронная фабрика
ООО Саратовская макаронная фабрика Фото-зоны. Декор
Фото-зоны. Декор Типы торговых точек
Типы торговых точек Manpower Russia & Region для СКБ-Контур
Manpower Russia & Region для СКБ-Контур Бренд Старый Мельник из Бочонка
Бренд Старый Мельник из Бочонка Люксоптика. Перша українська Компанія, яка об’єднала в собі здоров’я, стиль і красу
Люксоптика. Перша українська Компанія, яка об’єднала в собі здоров’я, стиль і красу Работа с контентом и создание контент-плана
Работа с контентом и создание контент-плана Проверка гипотез. Продукт для клиента
Проверка гипотез. Продукт для клиента Современный рынок мобильной рекламы в России и мире
Современный рынок мобильной рекламы в России и мире Сущность, содержание и инструментарий маркетинга
Сущность, содержание и инструментарий маркетинга Программа лояльности для питомников и заводчиков
Программа лояльности для питомников и заводчиков Лаборатория Деда Мороза. Программа для детей от 5-12 лет
Лаборатория Деда Мороза. Программа для детей от 5-12 лет Отели на о.Эвия. Эдипсос
Отели на о.Эвия. Эдипсос Приглашение на предквалификационный отбор. Доставка клиентам СПБ и ЛО, ноябрь 2023
Приглашение на предквалификационный отбор. Доставка клиентам СПБ и ЛО, ноябрь 2023