- Главная
- Без категории
- State diagram of single component system

Содержание
- 2. Component - chemically recognizable species (Fe and C in carbon steel, H2O and NaCl in salted
- 3. A phase diagram - graphical representation of the combinations of temperature, pressure, composition, or other variables
- 4. Interpretation of Phase Diagrams For a given temperature and composition we can use phase diagram to
- 18. Скачать презентацию
Слайд 2
Component - chemically recognizable species (Fe and C
in carbon steel, H2O
Component - chemically recognizable species (Fe and C
in carbon steel, H2O
and NaCl in salted water). A binary
alloy contains two components, a ternary alloy – three, etc.
Phase – a portion of a system that has uniform physical
and chemical characteristics. Two distinct phases in a
system have distinct physical or chemical characteristics
(e.g. water and ice) and are separated from each other by
definite phase boundaries. A phase may contain one or
more components.
A single-phase system is called homogeneous,
systems with two or more phases are mixtures or
heterogeneous systems.
D
alloy contains two components, a ternary alloy – three, etc.
Phase – a portion of a system that has uniform physical
and chemical characteristics. Two distinct phases in a
system have distinct physical or chemical characteristics
(e.g. water and ice) and are separated from each other by
definite phase boundaries. A phase may contain one or
more components.
A single-phase system is called homogeneous,
systems with two or more phases are mixtures or
heterogeneous systems.
D
Definitions: Components and Phases
Definitions: Components and Phases
Слайд 3
A phase diagram - graphical representation of the
combinations of temperature, pressure,
A phase diagram - graphical representation of the
combinations of temperature, pressure,
composition, or
other variables for which specific phases exist at
equilibrium.
For H2O, a typical diagram shows the temperature and
pressure at which ice (solid),water (liquid) and steam (gas)
Exis
other variables for which specific phases exist at
equilibrium.
For H2O, a typical diagram shows the temperature and
pressure at which ice (solid),water (liquid) and steam (gas)
Exis
Слайд 4
Interpretation of Phase Diagrams
For a given temperature and composition we can
Interpretation of Phase Diagrams
For a given temperature and composition we can
use phase
diagram to determine:
diagram to determine:
Finding the composition in a two phase region:
1. Locate composition and temperature in diagram
2. In two phase region draw the tie line or isotherm
3. Note intersection with phase boundaries. Read
compositions at the intersections.
The liquid and solid phases have these compositions.
Слайд 5
Слайд 6
Слайд 7
Слайд 8
Слайд 9
Слайд 10
Слайд 11
Слайд 12
Слайд 13
Слайд 14
Слайд 15
Слайд 16
- Предыдущая
Chemistry in our lifeСледующая -
Корпоративный стиль: функции и элементы










 Антисоциальные и криминальные молодежные группы
Антисоциальные и криминальные молодежные группы Флористика. Изготовление плоскостных композиций из засушенных цветов, листьев, лепестков
Флористика. Изготовление плоскостных композиций из засушенных цветов, листьев, лепестков 20230808_urok-sorevnovanie_8_fizika
20230808_urok-sorevnovanie_8_fizika Природа и население Европейского Севера
Природа и население Европейского Севера Робототехника. Fischertechnik - инструментарий для развития инженерных знаний и навыков учащихся, через игру
Робототехника. Fischertechnik - инструментарий для развития инженерных знаний и навыков учащихся, через игру Русская народная сказка Петух и собака. Урок чтения, 1 класс
Русская народная сказка Петух и собака. Урок чтения, 1 класс Лабораторные работы и методические рекомендации
Лабораторные работы и методические рекомендации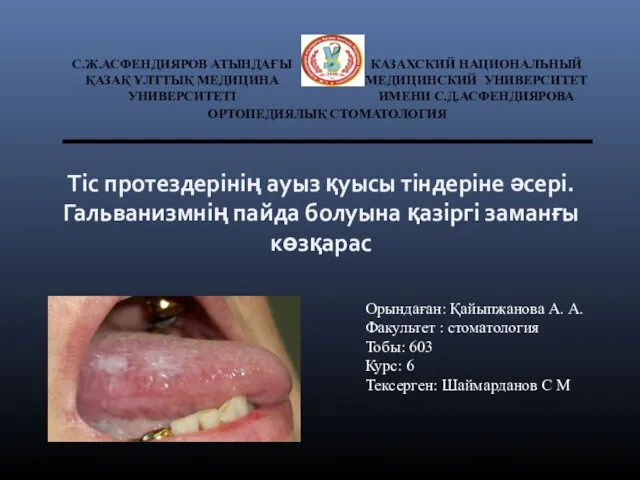 Тіс протездерін көтере алмау себептері
Тіс протездерін көтере алмау себептері Қазақстан республикасының мұнай – газ саласының қазіргі жағдайы
Қазақстан республикасының мұнай – газ саласының қазіргі жағдайы Презентация Правовое воспитание
Презентация Правовое воспитание Культура Беларуси XIV-1п.XVI вв
Культура Беларуси XIV-1п.XVI вв Dark wash-Finish
Dark wash-Finish Запуск стартапов и новые форматы школ для предпринимателей
Запуск стартапов и новые форматы школ для предпринимателей Производство колбас и колбасных изделий
Производство колбас и колбасных изделий Пояснительная записка. Южная специализированная производственная база БЕ 2901
Пояснительная записка. Южная специализированная производственная база БЕ 2901 Опыт работы по теме Деятельность классного руководителя по формированию нравственного поведения младших школьников
Опыт работы по теме Деятельность классного руководителя по формированию нравственного поведения младших школьников Портфолио преподавателя информатики
Портфолио преподавателя информатики Термическая обработка сварных изделий
Термическая обработка сварных изделий Грозова енергетика
Грозова енергетика Тромболитическая терапия
Тромболитическая терапия Праздник Пасха.
Праздник Пасха. If I had a million dollars
If I had a million dollars День 23 февраля
День 23 февраля Довольствование или довольство. Библейское основание управления финансами
Довольствование или довольство. Библейское основание управления финансами Стратегическая сессия ГК Гео-проект
Стратегическая сессия ГК Гео-проект Аппликация, виды аппликации
Аппликация, виды аппликации Затратный подход к оценке собственности
Затратный подход к оценке собственности Сім чудес України
Сім чудес України