Содержание
- 2. LECTURE OBJECTIVES: After reading this lesson, the learner will be able to state the postulates of
- 3. Skills to Develop To know the most common structures observed for metal complexes. To predict the
- 4. A salt is formed by the neutralization of an acid by a base. There are different
- 5. One of the important aspects of any chemical research is the preparation of new compounds. This
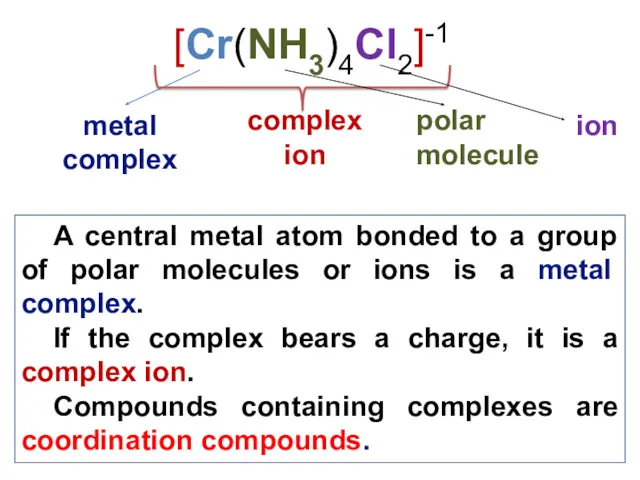
- 7. A central metal atom bonded to a group of polar molecules or ions is a metal
- 8. Coordination complexes (a complex ion) is comprised of two important parts: the central atom and its
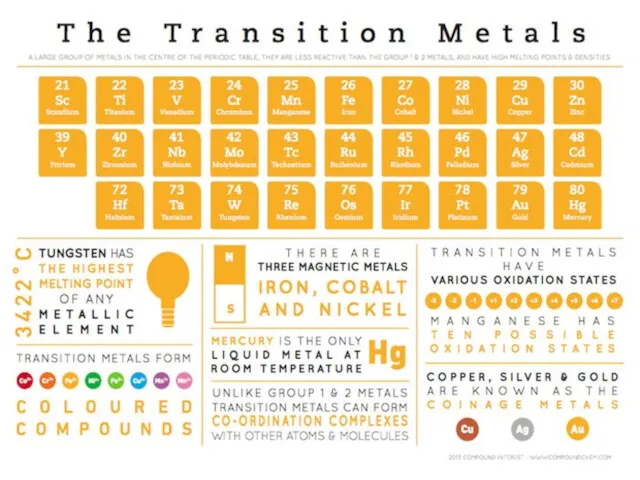
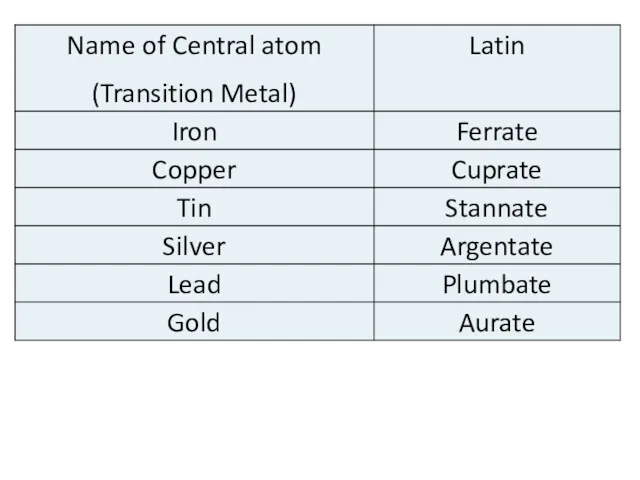
- 9. THE CENTRAL ATOM It is can be: any metallic ion (usually a transition d- or f-metal);
- 10. Coordination compounds are complex or contain complex ions, for example: Complex Cation: [Co(NH3)6]3+ Complex Anion: [CoCl4(NH3)2]−1
- 11. The modern theory of coordination chemistry is based largely on the work of Alfred Werner (1866–1919;
- 12. STRUCTURE OF COMPLEXES: Central Metal Ion: It is an acceptor atom containing vacant orbitals to which
- 13. Coordination Sphere: Central atom and ligands comprise the inner coordination sphere; complex ion enclosed in square
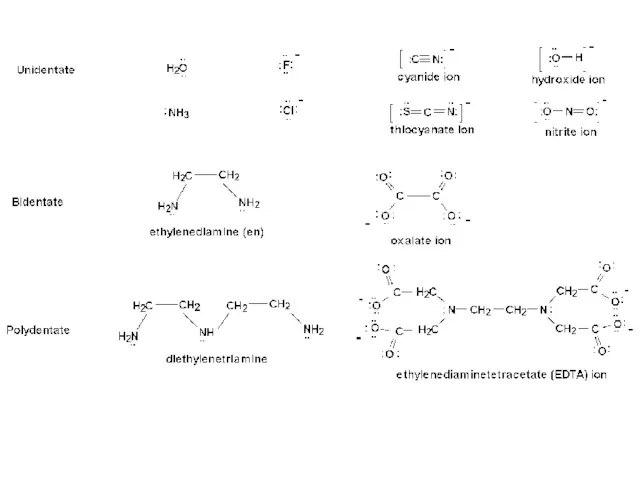
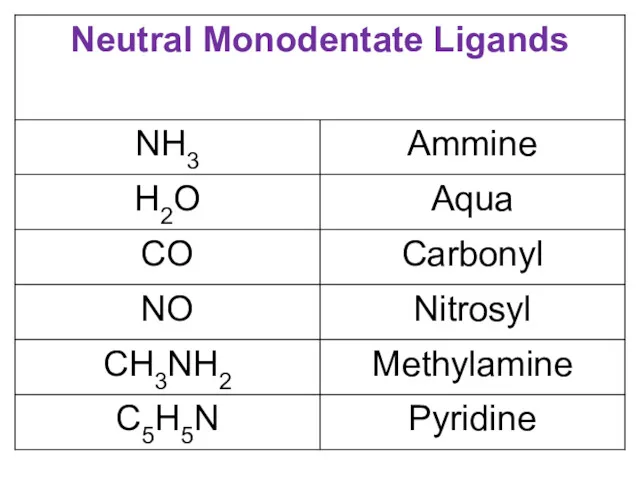
- 14. LIGANDS It is an ion or polar molecule capable of donating a pair of electrons to
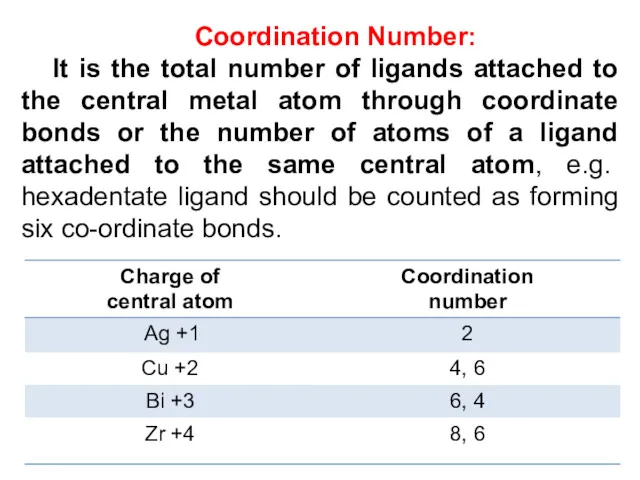
- 16. Coordination Number: It is the total number of ligands attached to the central metal atom through
- 17. Oxidation number: It is the charge which the central atom appears to have if all the
- 18. Or, knowing the oxidation number on the metal and the charges on the ligands, one can
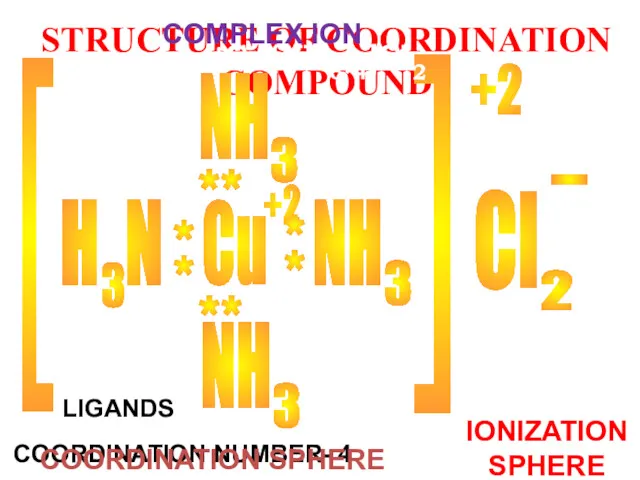
- 19. STRUCTURE OF COORDINATION COMPOUND Cu Cl +2 - [ [ [Cu(NH3)4]Cl2 КОМПЛЕКСООБРАЗОВАТЕЛЬ LIGANDS COORDINATION NUMBER- 4
- 20. Classification of complex compounds: By the charge of the complex ion [Cr(H2O)4]3+Cl3 [PtCl4(NH3)2] 0 K2[PtCl6]2- [Cu(NH3)4]2+[PtCl4]2-
- 21. By composition of ionization sphere H2[PtCl6] Na3[AlF6] [Pt(NH3)2Cl2] [Ag(NH3)2]OH
- 22. By the nature of the ligand: [Fe(H2O)6]SO4 K[Au(CN)4] [Zn(NH3)4]Cl2 [CoCl(NH3)3(H2O)2](NO3)2
- 23. Nomenclature of Coordination Compounds The basic protocol in coordination nomenclature is to name the ligands attached
- 27. Nomenclature of Coordination Compounds As is the case with ionic compounds, the name of the cation
- 28. Nomenclature of Coordination Compounds The names of anionic ligands end in “o”; the endings of the
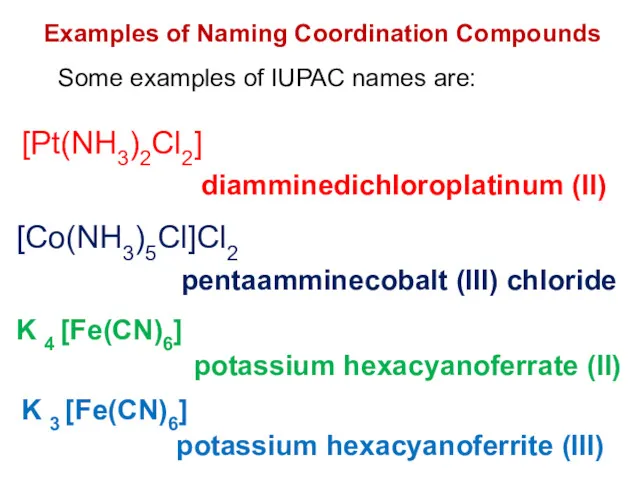
- 29. Examples of Naming Coordination Compounds Some examples of IUPAC names are: diamminedichloroplatinum (II) pentaamminecobalt (III) chloride
- 30. Preparation of complex compounds Complex compounds prepared by such reactions: 1) Connection reactions: HgI2 + 2KI
- 31. Nature of chemical bonding in complex compounds Structure physico-chemical and biological properties of complex compounds depend
- 32. PROPERTIES OF COMPLEX COMPOUNDS 1) Dissociation of complex compounds. Complex compounds are strong electrolytes. In aqueous
- 33. 2) Stability of complex compounds. This formation constant (Kf), describes the formation of a complex ion
- 35. Скачать презентацию













![Classification of complex compounds: By the charge of the complex ion [Cr(H2O)4]3+Cl3 [PtCl4(NH3)2] 0 K2[PtCl6]2- [Cu(NH3)4]2+[PtCl4]2-](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/142977/slide-19.jpg)
![By composition of ionization sphere H2[PtCl6] Na3[AlF6] [Pt(NH3)2Cl2] [Ag(NH3)2]OH](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/142977/slide-20.jpg)
![By the nature of the ligand: [Fe(H2O)6]SO4 K[Au(CN)4] [Zn(NH3)4]Cl2 [CoCl(NH3)3(H2O)2](NO3)2](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/142977/slide-21.jpg)








 Валентность элементов. Определение валентности по формулам
Валентность элементов. Определение валентности по формулам Электролиз Урок для 11 класса
Электролиз Урок для 11 класса Двойной электрический слой. Теория Гельмгольца
Двойной электрический слой. Теория Гельмгольца Расчет процесса горения: состав горючей системы и расчет количества воздуха, необходимого для горения
Расчет процесса горения: состав горючей системы и расчет количества воздуха, необходимого для горения Атомы, молекулы и ионы. Вещества молекулярного и немолекулярного строения
Атомы, молекулы и ионы. Вещества молекулярного и немолекулярного строения Летучие яды. Токсичные газы
Летучие яды. Токсичные газы Минералдар
Минералдар Химическая кинетика
Химическая кинетика Периодическая система Д. И. Менделеева
Периодическая система Д. И. Менделеева Гидролиз солей
Гидролиз солей Спирти. Загальні відомості про спирти
Спирти. Загальні відомості про спирти Силикат өнеркəсібі
Силикат өнеркəсібі Химические реакторы
Химические реакторы Закон Авогадро. Молярный объем газов
Закон Авогадро. Молярный объем газов Йод. Йодтың ашылуы
Йод. Йодтың ашылуы Свойства воды
Свойства воды Химия элементов. Комплексные соединения. Основные понятия координационной теории. Номенклатура. Поведение в растворе
Химия элементов. Комплексные соединения. Основные понятия координационной теории. Номенклатура. Поведение в растворе Новинки СН старт: июль 2016 Профессиональная химия PRO service
Новинки СН старт: июль 2016 Профессиональная химия PRO service Химия и современный быт человека
Химия и современный быт человека Классификация химических элементов. 8 класс
Классификация химических элементов. 8 класс Вещества. Чистые вещества. Смеси
Вещества. Чистые вещества. Смеси Углеводороды. Предельные (насыщенные) углеводороды
Углеводороды. Предельные (насыщенные) углеводороды Р-элементы IV А группы. Углерод и кремний
Р-элементы IV А группы. Углерод и кремний Химия в продуктах питания
Химия в продуктах питания Подготовка к ГИА. А14. Определение характера среды раствора кислот и щелочей с помощью индикаторов. Реакции на ионы в растворе
Подготовка к ГИА. А14. Определение характера среды раствора кислот и щелочей с помощью индикаторов. Реакции на ионы в растворе Синтез высокомолекулярных соединений
Синтез высокомолекулярных соединений Геохимия
Геохимия Проектная деятельность школьников в процессе обучения химии
Проектная деятельность школьников в процессе обучения химии