Содержание
- 2. in analytical chemistry; for allocation of chemical elements; in galvanotechnics; for corrosion control; in the production
- 3. According to their contents, chemicals are divided into simple substances (H2, Cl2, O2 etc) and compounds
- 4. According to this theory, in each complex compound there is a сentral ion (complexing), other ions,
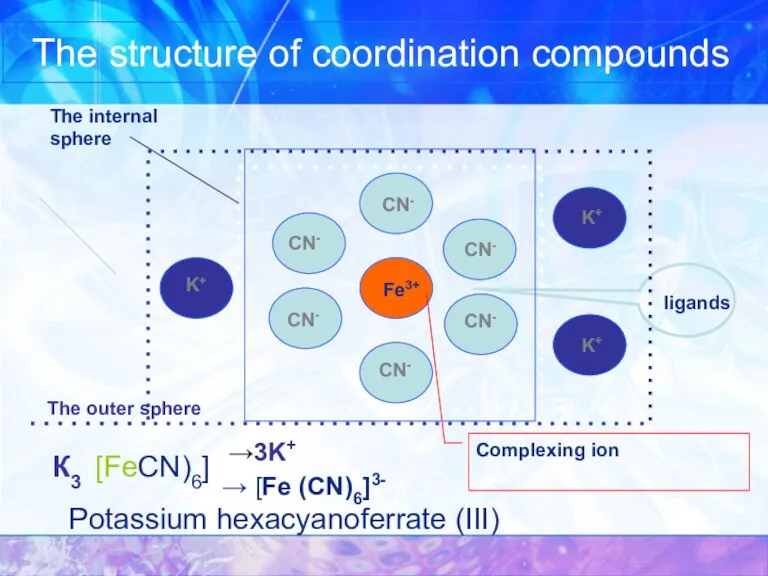
- 5. The structure of coordination compounds Potassium hexacyanoferrate (III) Fe3+ CN- CN- CN- CN- CN- CN- K+
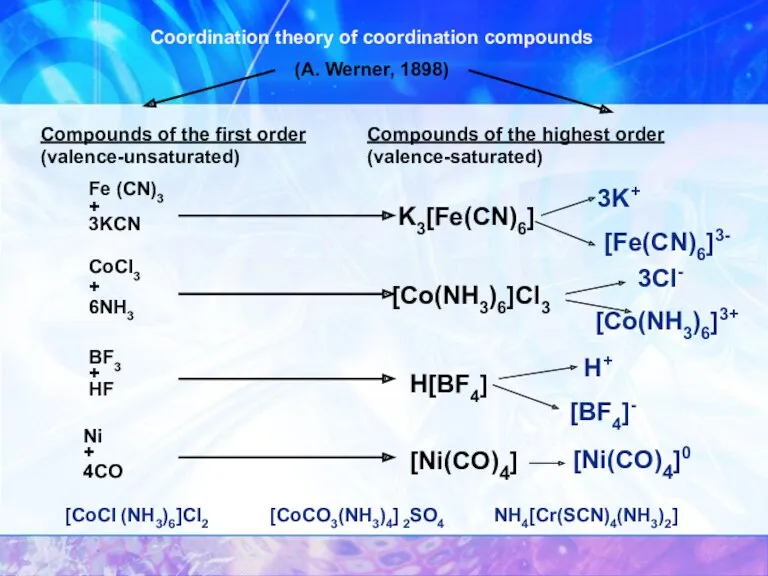
- 6. Coordination theory of coordination compounds (A. Werner, 1898) Compounds of the first order (valence-unsaturated) Compounds of
- 7. Complex compounds : Coordination compounds (complexing ion+ ligands) Molecular complexes: Quinone-hydroquinone; Hydroquinone-methanol Inclusion compounds: 6Н2О*X, where
- 8. 2. Classification and nomenclature of coordination compounds Cation is named at first, then anion. Name of
- 9. Nomenclature of coordinational compounds К[Co(CN)4(CO)2] - potassium dicarbonyltetracyanocobaltate(ІІІ) 1) NH4SCN + Cr(SCN)3 + 2NH3 NH4[Cr(SCN)4(NH3)2] ammonium
- 10. Classification of coordination compounds on different grounds 1. According to the sign of the electric charge
- 11. 2. According to their attachment to the definite class of compounds complex diamminesilver(I) hydroxydum acids bases
- 12. 3. By the nature of the ligands Ammines [Ni(NH3)6]Cl2 Aquacomplexes [Co(H2O)6]SO4 Acidocomplexes K[Cu(CN)2] Карбонилы [Fe(CO)5] Hydroxocomplexes
- 13. 4. По внутренней структуре комплексного соединения Моноядерные 4.2. По наличию циклов 4.1. По числу ядер Полиядерные
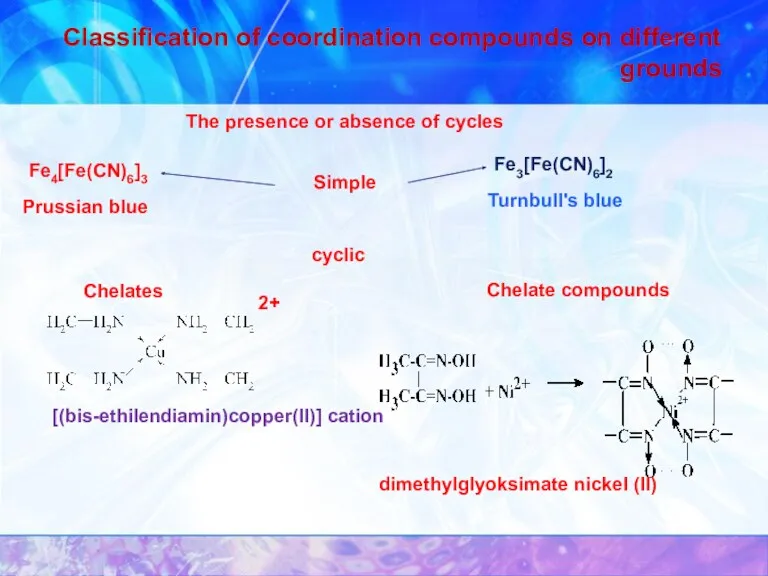
- 14. Classification of coordination compounds on different grounds The presence or absence of cycles Simple Fe4[Fe(CN)6]3 Prussian
- 15. Isomerism of coordination compounds Coordination isomerism is different allocation of ligand in internal coordination spheres. Interact
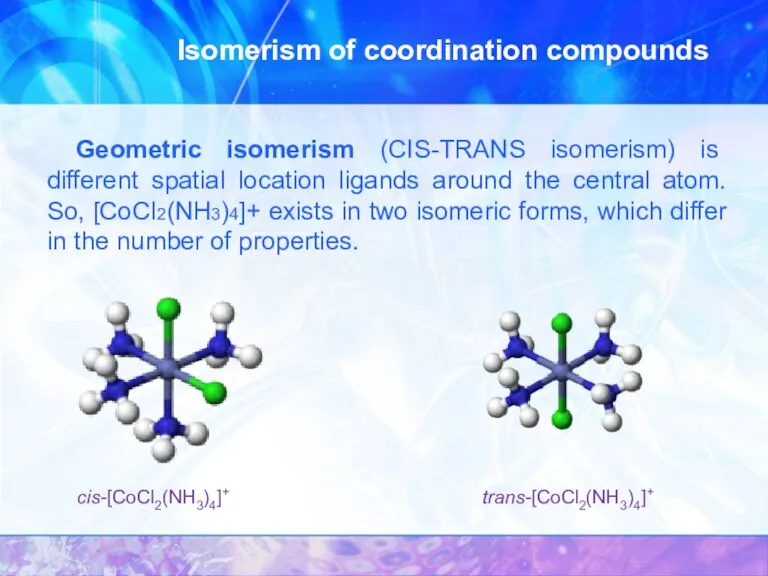
- 16. Isomerism of coordination compounds Geometric isomerism (CIS-TRANS isomerism) is different spatial location ligands around the central
- 17. Isomerism of coordination compounds Optical isomerism. Optical isomerism characterized by the ability to rotate the plane
- 18. Isomerism of coordination compounds Structural isomerism Hydrated (solvation) isomerism is different allocation of molecules of the
- 19. [Co(H2O)6]2+ pink [Co(CH3COO)2] hot pink [Co(NO2)6]4- orange [Co(NH3)6]2+ brownish-pink Amplification of a field of ligands Influence
- 20. 2. The reactions of coordination compounds. Stability of complex compounds and constant instability.
- 21. K3[Fe(CN)6] 3K+ + [Fe(CN)6]3- [Ag(NH3)2]Cl [Ag(NH3)2]+ + Cl- Dissociation of the CC on the external sphere
- 22. Reactions of complex on the outer sphere 2K3[Fe(CN)6] + 3FeSO4 = Fe3[Fe(CN)6]2↓ + 3K2SO4 [CoCl2(NH3)4]Cl +
- 23. Br- Br- 2Br- Cu2+ [CuBr]+ [CuBr2] [CuBr4]2- + H2O + H2O + H2O Stepped formation and
- 24. Complexing constants Joining the ligand L to the ion (atom) - M complexing to form a
- 25. [Ag(NH3)2]+ [Ag(NH3)]+ + NH3 [Ag(NH3)2]+ Ag+ + 2 NH3 Dissociation of the CC the inner sphere
- 26. At the same coordination number Comparison of the strength of the complexes according general instability constants
- 27. With different coordination number
- 28. With different coordination number 3. Comparison of the stability of the complexes of the stepped constants
- 29. The reaction of complexes with the destruction of the complex 1. The formation of the stronger
- 30. 3. Dilution K[AgCl2] = KCl + AgCl↓ 5. Red-ox reactions 2K3[Cr(ОH)6] + 3Сl2 + 4KOH =
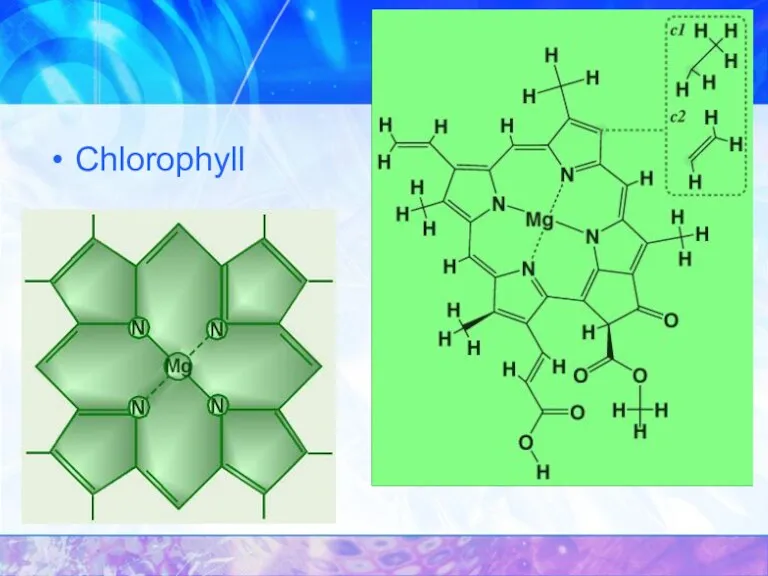
- 31. Chlorophyll
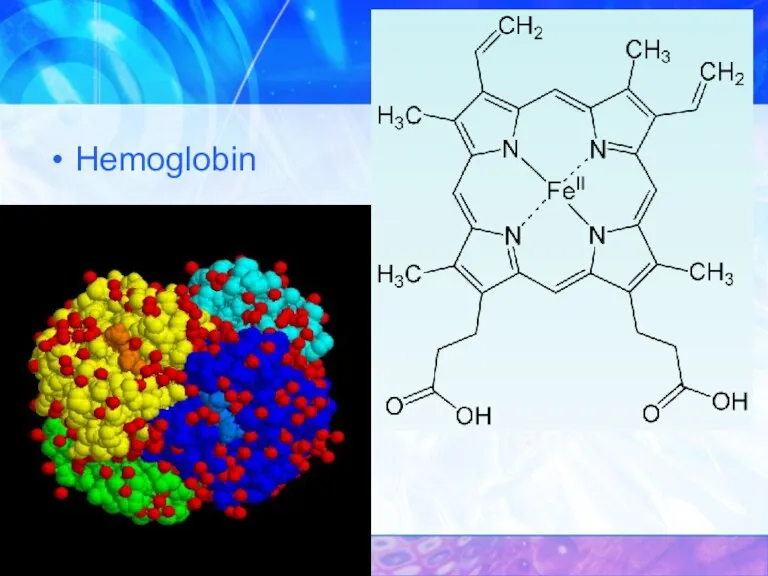
- 32. Hemoglobin
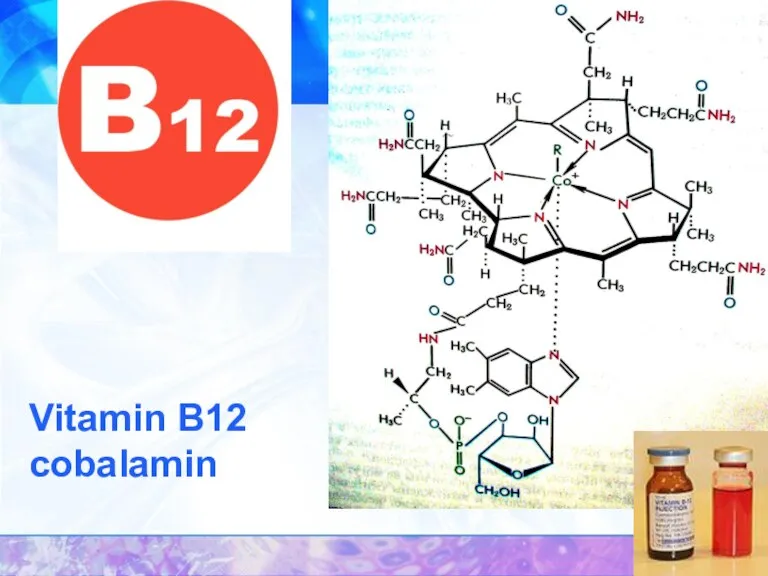
- 33. Vitamin B12 cobalamin
- 34. THE APPLICATION OF COMPLEXES IN MEDICINE Substances, eliminating effects of poisons on the biological structure and
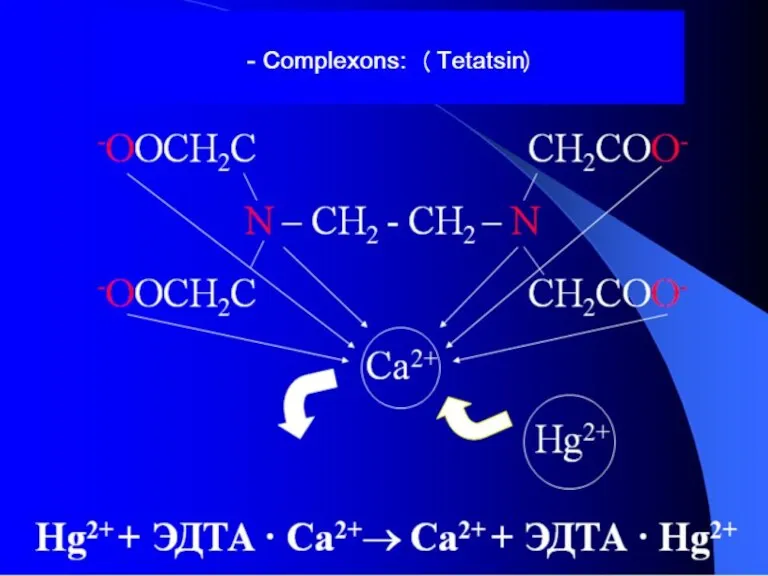
- 35. -antidotes: (Dimercaprol) EDTA, Na2EDTA, Na2CaEDTA Mercaptide bond
- 36. THE APPLICATION OF COMPLEXES IN MEDICINE For the lead removing using preparations based on EDTA. The
- 38. -anticancer drug: dihlorodiamminplatinum cis-isomer (cis-platin) cis- [Pt(NH3)2Cl2] cis- [Pt(NH3)4Cl2]
- 40. Скачать презентацию





![Nomenclature of coordinational compounds К[Co(CN)4(CO)2] - potassium dicarbonyltetracyanocobaltate(ІІІ) 1) NH4SCN](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-8.jpg)


![3. By the nature of the ligands Ammines [Ni(NH3)6]Cl2 Aquacomplexes](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-11.jpg)




![[Co(H2O)6]2+ pink [Co(CH3COO)2] hot pink [Co(NO2)6]4- orange [Co(NH3)6]2+ brownish-pink Amplification](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-18.jpg)

![K3[Fe(CN)6] 3K+ + [Fe(CN)6]3- [Ag(NH3)2]Cl [Ag(NH3)2]+ + Cl- Dissociation of](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-20.jpg)
![Reactions of complex on the outer sphere 2K3[Fe(CN)6] + 3FeSO4](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-21.jpg)
![Br- Br- 2Br- Cu2+ [CuBr]+ [CuBr2] [CuBr4]2- + H2O +](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-22.jpg)

![[Ag(NH3)2]+ [Ag(NH3)]+ + NH3 [Ag(NH3)2]+ Ag+ + 2 NH3 Dissociation](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-24.jpg)




![3. Dilution K[AgCl2] = KCl + AgCl↓ 5. Red-ox reactions](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-29.jpg)



![-anticancer drug: dihlorodiamminplatinum cis-isomer (cis-platin) cis- [Pt(NH3)2Cl2] cis- [Pt(NH3)4Cl2]](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/441733/slide-37.jpg)
 Crystal structure and surface phase composition of palladium oxides thin films for gas sensors
Crystal structure and surface phase composition of palladium oxides thin films for gas sensors Моноядерні арени
Моноядерні арени Катенаны. Ротаксаны (лекция 6)
Катенаны. Ротаксаны (лекция 6) Вуглеводи
Вуглеводи Коррозия металлов и способы защиты от неё. (11 класс)
Коррозия металлов и способы защиты от неё. (11 класс) Периодический закон Д.И. Менделеева
Периодический закон Д.И. Менделеева Классы неорганических веществ. Лекция №2
Классы неорганических веществ. Лекция №2 Сопряжение. Ароматичность. Электронные эффекты заместителей
Сопряжение. Ароматичность. Электронные эффекты заместителей Наноструктурные и нанокристаллические материалы
Наноструктурные и нанокристаллические материалы Аммиак. Состав вещества
Аммиак. Состав вещества Азотная кислота и её соли
Азотная кислота и её соли Ауыз судың химиялық тұрғыдан зиянсыздығын сипаттаушы көрсеткіштер
Ауыз судың химиялық тұрғыдан зиянсыздығын сипаттаушы көрсеткіштер КИСЛОРОД
КИСЛОРОД Воздух и его состав. Урок-презентация. 8 класс
Воздух и его состав. Урок-презентация. 8 класс Алкени і алкіни
Алкени і алкіни История хроматографии. Виды хроматографических методов
История хроматографии. Виды хроматографических методов Генетичні зв’язки між неорганічними речовинами. Практична робота 2
Генетичні зв’язки між неорганічними речовинами. Практична робота 2 Положение кислорода в периодической системе. Электронное строение
Положение кислорода в периодической системе. Электронное строение Фосфор
Фосфор Химия и здоровье
Химия и здоровье Камни и Скорпион
Камни и Скорпион Полимеры. Протезы трахеи
Полимеры. Протезы трахеи Облагораживание тяжелых нефтей и природных битумов
Облагораживание тяжелых нефтей и природных битумов Кристалогідрати. Номенклатура, найважливіші кристалогідрати та їх застосування
Кристалогідрати. Номенклатура, найважливіші кристалогідрати та їх застосування Спирты. Физические и химические свойства
Спирты. Физические и химические свойства Компоненты, попадающие в продукты питания из минеральных и других удобрений
Компоненты, попадающие в продукты питания из минеральных и других удобрений Серная кислота. Занимательные опыты
Серная кислота. Занимательные опыты Гидролиз органических и неорганических соединений
Гидролиз органических и неорганических соединений