Содержание
- 2. Physical and Chemical Properties of Oil Oil is described by the physical properties of density, color,
- 3. Colour Petroleum can be of different colors. Oil colors vary in a very wide range from
- 4. Density The SI unit is kg\ m3 at a reference temperature, typically 15 °C. Knowledge of
- 5. Density 5
- 6. Viscosity Viscosity is a property of fluids that indicates their resistance to flow, defined as ratio
- 7. Viscosity 7
- 8. Thermal expansion The degree of expansion is expressed as the coefficient of thermal expansion. Thermal expansion
- 9. Thermal expansion 9
- 10. Crude oil Crude oils are complex mixtures containing hundreds of different hydrocarbon compounds that vary in
- 11. Crude oil 11
- 12. Crude oil All petroleum hydrocarbons are divided into three groups: Alkanes (methane group) with the general
- 14. Скачать презентацию
Слайд 2
Physical and Chemical Properties of Oil
Oil is described by the physical
Physical and Chemical Properties of Oil
Oil is described by the physical
properties of density, color, viscosity, thermal expansion and other properties related to the number of carbon atoms in the molecules.
2
Слайд 3
Colour
Petroleum can be of different colors. Oil colors vary in a
Colour
Petroleum can be of different colors. Oil colors vary in a
very wide range from oilfield to oilfield: from pale yellow, yellow and even colourless to dark grey, green and dark brown shades.
3
Слайд 4
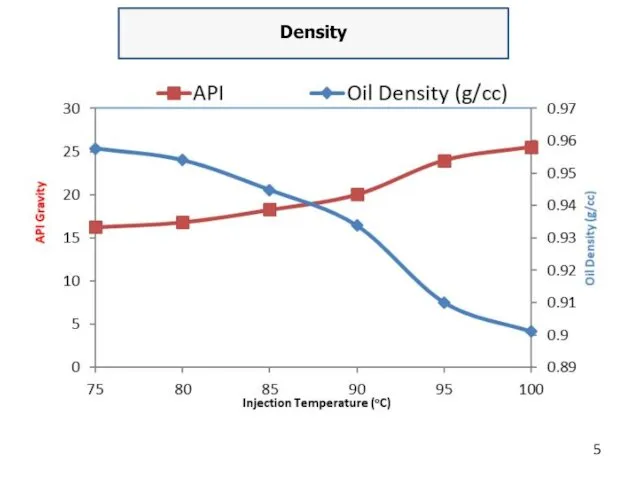
Density
The SI unit is kg\ m3 at a reference temperature, typically
Density
The SI unit is kg\ m3 at a reference temperature, typically
15 °C. Knowledge of density is required for quantity calculations. In the USA and some other countries the density of petroleum products is defined in terms of API gravity. This is an arbitrary scale adopted by the American Petroleum Institute for expressing the relative density of oils. The API gravity scale is read "backwards". The higher the API number, expressed as degrees API, the less dense (lighter) the oil is. Conversely, the lower the degrees API, the more dense (heavier) is the oil.
Density of oils range from 0.65 to 1.0 gr\cm3 and more at 20 °C. According to density, oils may be light, medium and heavy. Light oil is characterized by the density of 0.5–0.87; medium oil: 0.871–0.910 and heavy oil is described being as 0.910–1.05 gr\cm3.
Density of oils range from 0.65 to 1.0 gr\cm3 and more at 20 °C. According to density, oils may be light, medium and heavy. Light oil is characterized by the density of 0.5–0.87; medium oil: 0.871–0.910 and heavy oil is described being as 0.910–1.05 gr\cm3.
4
Слайд 5
Density
5
Density
5
Слайд 6
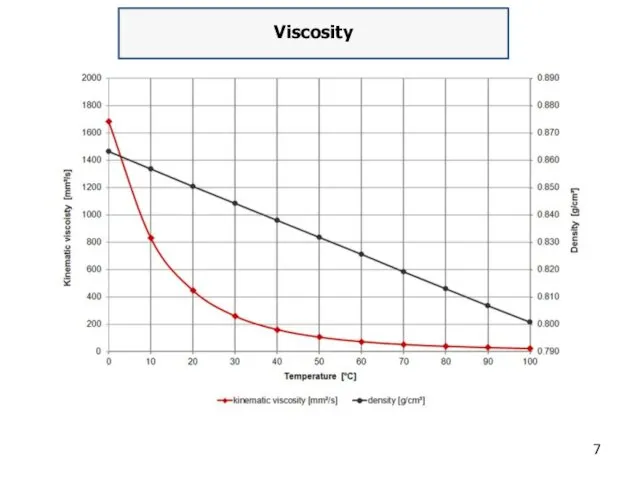
Viscosity
Viscosity is a property of fluids that indicates their resistance to
Viscosity
Viscosity is a property of fluids that indicates their resistance to
flow, defined as ratio of shear stress to shear rate. Crude oils range in consistency from water-like to tar-like solids. Fluid with a high viscosity such as syrup deforms more slowly than fluid with a low viscosity such as water. Absolute viscosity is measured in Poise.
The oil specific viscosity is usually defined as ratio of absolute viscosity of a given fluid to absolute viscosity of water at the same temperature. The viscosity of oil is dependent upon temperature, pressure and shear rate. Viscosity decreases as temperature increases because molecules vibrate and interact less.
Conversely, the viscosity of oil increases as temperature decreases and it can become grease-like at very low temperature. The volume of given oil mass increases with temperature, therefore, its density decreases.
The oil specific viscosity is usually defined as ratio of absolute viscosity of a given fluid to absolute viscosity of water at the same temperature. The viscosity of oil is dependent upon temperature, pressure and shear rate. Viscosity decreases as temperature increases because molecules vibrate and interact less.
Conversely, the viscosity of oil increases as temperature decreases and it can become grease-like at very low temperature. The volume of given oil mass increases with temperature, therefore, its density decreases.
6
Слайд 7
Viscosity
7
Viscosity
7
Слайд 8
Thermal expansion
The degree of expansion is expressed as the coefficient of
Thermal expansion
The degree of expansion is expressed as the coefficient of
thermal expansion. Thermal expansion is useful to determine the size of container needed when the oil is heated. Thermal expansion is expressed as the ratio of volume change to initial volume after heating 10 °C.
8
Слайд 9
Thermal expansion
9
Thermal expansion
9
Слайд 10
Crude oil
Crude oils are complex mixtures containing hundreds of different hydrocarbon
Crude oil
Crude oils are complex mixtures containing hundreds of different hydrocarbon
compounds that vary in appearance and composition from oil field to oil field, therefore, in various oil fields the oil composition can vary significantly.
All hydrocarbons are divided into two groups: saturated hydrocarbons and unsaturated hydrocarbons. Saturated hydrocarbons are not capable of attaching atoms and molecules while unsaturated hydrocarbons are capable of attaching atoms and molecules. The latter take part in chemical reactions easier. Hydrocarbons can be as simple as methane, but many are highly complex molecules and can occur as gases, liquids or solids.
An "average" crude oil contains about 84 % carbon, 14 % hydrogen, 1–5 % sulfur, and less than 1 % of nitrogen, oxygen, metals and salts. In the refinery, most of these non-hydrocarbon substances are removed and the oil is broken down into various compounds and blended into useful products.
All hydrocarbons are divided into two groups: saturated hydrocarbons and unsaturated hydrocarbons. Saturated hydrocarbons are not capable of attaching atoms and molecules while unsaturated hydrocarbons are capable of attaching atoms and molecules. The latter take part in chemical reactions easier. Hydrocarbons can be as simple as methane, but many are highly complex molecules and can occur as gases, liquids or solids.
An "average" crude oil contains about 84 % carbon, 14 % hydrogen, 1–5 % sulfur, and less than 1 % of nitrogen, oxygen, metals and salts. In the refinery, most of these non-hydrocarbon substances are removed and the oil is broken down into various compounds and blended into useful products.
10
Слайд 11
Crude oil
11
Crude oil
11
Слайд 12
Crude oil
All petroleum hydrocarbons are divided into three groups:
Alkanes (methane group)
Crude oil
All petroleum hydrocarbons are divided into three groups:
Alkanes (methane group)
with the general formula CnH2n + 2. This group represents saturated hydrocarbons, since all their valence bonds are involved. From a chemical point of view, they are the most inert, in other words, unable to react with other chemical compounds. The structure of alkanes can be either linear (normal alkanes) or branched (isoalkanes).
Cyclanes (naphthenic group) with the general formula CnH2n. Their main attribute is a five- or six-membered ring consisting of carbon atoms. In other words, cyclanes, unlike alkanes, have a cyclic structure closed in a chain. This group also represents the limiting (saturated) compounds, and they also hardly enter into reactions with other chemical elements.
Arenas (aromatic group) with the general formula CnH2n-6. Their structure is six-membered cycles, which are based on the aromatic benzene core (C6H6). They are distinguished by the presence of double bonds between the atoms. Arenas are monocyclic (one benzene ring), bicyclic (double benzene rings) and polycyclic (rings are connected according to the principle of honeycombs).
Cyclanes (naphthenic group) with the general formula CnH2n. Their main attribute is a five- or six-membered ring consisting of carbon atoms. In other words, cyclanes, unlike alkanes, have a cyclic structure closed in a chain. This group also represents the limiting (saturated) compounds, and they also hardly enter into reactions with other chemical elements.
Arenas (aromatic group) with the general formula CnH2n-6. Their structure is six-membered cycles, which are based on the aromatic benzene core (C6H6). They are distinguished by the presence of double bonds between the atoms. Arenas are monocyclic (one benzene ring), bicyclic (double benzene rings) and polycyclic (rings are connected according to the principle of honeycombs).
12
- Предыдущая
Совместная работа трёх братьев мастеров






 Основи. Властивості, застосування гідроксидів Натрію і Калію
Основи. Властивості, застосування гідроксидів Натрію і Калію Методи добування у промисловості
Методи добування у промисловості Арены - ароматические соединения
Арены - ароматические соединения Щелочные металлы и их соединения. 9 класс
Щелочные металлы и их соединения. 9 класс Кремний. Физические свойства кремния
Кремний. Физические свойства кремния Аспирин: польза или вред?
Аспирин: польза или вред? Щелочные металлы
Щелочные металлы Углерод, как химический элемент и простое вещество
Углерод, как химический элемент и простое вещество Химический состав воздуха
Химический состав воздуха Оптические свойства дисперсных систем. Оптические методы исследования коллоидных систем
Оптические свойства дисперсных систем. Оптические методы исследования коллоидных систем Алканы (Предельные углеводороды. Парафины. Насыщенные углеводороды)
Алканы (Предельные углеводороды. Парафины. Насыщенные углеводороды) Реакции ионного обмена в растворах электролитов. рH раствора как показатель кислотности среды
Реакции ионного обмена в растворах электролитов. рH раствора как показатель кислотности среды Germaniy gruppachasining elementlarining olinishi va xossalari
Germaniy gruppachasining elementlarining olinishi va xossalari Фосфор и его соединения
Фосфор и его соединения Водород
Водород Вода — это уникальное вещество
Вода — это уникальное вещество Скорость химических реакций. Факторы, влияющие на скорость химической реакции
Скорость химических реакций. Факторы, влияющие на скорость химической реакции Биополимеры. Классификация полисахаридов (гликаны)
Биополимеры. Классификация полисахаридов (гликаны) Минералы и формы существования марганца
Минералы и формы существования марганца Экспериментальные методы физико-химических исследований. Лекция 7
Экспериментальные методы физико-химических исследований. Лекция 7 Щелочные металлы
Щелочные металлы Обобщающий урок по теме Металлы
Обобщающий урок по теме Металлы Альдегиды и кетоны
Альдегиды и кетоны Биологически важные вещества жиры
Биологически важные вещества жиры Виды химической связи
Виды химической связи Алкалоиды. Классификация алкалоидов:
Алкалоиды. Классификация алкалоидов: Разбор заданий #ОВРновый, #реакционно и #соточка
Разбор заданий #ОВРновый, #реакционно и #соточка Общие правила техники безопасности при работе в кабинете химии. Урок №2. Практическая работа №1
Общие правила техники безопасности при работе в кабинете химии. Урок №2. Практическая работа №1