Содержание
- 3. Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom or a
- 4. ЭЛЕКТРООТРИЦАТЕЛЬНОСТЬ ЭЛЕМЕНТОВ это способность одних атомов оттягивать на себя электроны от других атомов при взаимодействии
- 5. Not all atoms attract electrons with the same force. The amount of "pull" an atom exerts
- 6. An amount of electrons which was lost or gained by the atom in a chemical compound
- 8. The increase in oxidation state of an atom through a chemical reaction is known as an
- 9. In 1st reaction no change of oxidation degrees of atoms: In 2nd equation we see that
- 10. Oxidation-Reduction Reactions are all reactions that involve the change of an oxidation number, and transfer of
- 11. The chemical substance which loses electrons, is oxidised (as result increases its valency) and called the
- 12. IMPOTANT OXIDANTS An oxidizing agent: contains an element whose oxidation state decreases in a redox reaction
- 13. IMPOTANT REDUCTANTS A reducing agent: contains an element whose oxidation state increases in a redox reation
- 14. Some substances containing elements in the intermediate oxidation may be oxidants or reductants: Halogens in basic
- 15. H2+O2– Na2S+4O3 S0 H2+O–2 2О– +2е→2О-2 2О– -2е→ О20 Na2S–2 О20 Na2S+6O4 S+4 +6e → S–2
- 16. + 1 - 1 - 2 - 3 - 4 - 5 - 6 + 2
- 17. REDOX REACTION TYPES Intermolecular redox reactions in which the oxidant and reductant are part of different
- 18. DISPROPORTIONATION REACTIONS In some redox reactions, called disproportionation reactions, the same substance is both oxidized and
- 19. +5 +7 -1 -2е +6е 0 -1 +1 -1е +1е DISPROPORTIONATION REACTIONS
- 20. METHODS OF BALANCING REDOX REACTIONS 1) The Oxidation-Number Method (ionic balance): the key to this method
- 21. 2) Half Reactions Method (electronic balance): 1) Write and balance separate half-equations for oxidation and reduction,
- 22. H2O (+3e) KMnO4 Mn+2 MnO2 K2MnO4 H+ (+5e) OH– (+1e) In acidic solution: 2KMnO4 + 5Na2SO3+
- 23. Влияние среды на изменение степеней окисления атомов химических элементов
- 24. Влияние среды на изменение степеней окисления атомов химических элементов
- 25. Влияние среды на изменение степеней окисления атомов химических элементов
- 26. GLOSSARY Oxidation Any chemical change in which at least one element loses electrons, either completely or
- 28. Скачать презентацию
 Закон сохранения массы веществ. Химические уравнения
Закон сохранения массы веществ. Химические уравнения Полисахариды. Крахмал Целлюлоза. 10 класс
Полисахариды. Крахмал Целлюлоза. 10 класс Кислоты. 8 класс
Кислоты. 8 класс Химический состав нефтей. Ненасыщенные углеводороды. Алкены и алкадиены
Химический состав нефтей. Ненасыщенные углеводороды. Алкены и алкадиены Гідроліз солей
Гідроліз солей Введение в органическую химию
Введение в органическую химию Способы выражения концентрации в титриметрическом анализе
Способы выражения концентрации в титриметрическом анализе Алкалоиды и принципы их классификации
Алкалоиды и принципы их классификации Сложные эфиры
Сложные эфиры Розв’язування експериментальних задач (9 клас)
Розв’язування експериментальних задач (9 клас) Получение Н2, О2, щелочей
Получение Н2, О2, щелочей Chemical kinetics
Chemical kinetics Отрицательное влияние азотных минеральных удобрений на почву
Отрицательное влияние азотных минеральных удобрений на почву Жиры. Липиды и липоиды
Жиры. Липиды и липоиды Тағамның стериндері мен талшықтық құрамы
Тағамның стериндері мен талшықтық құрамы Iron Oxide
Iron Oxide Химические уравнения. Закон сохранения массы веществ
Химические уравнения. Закон сохранения массы веществ Альдегиды и кетоны
Альдегиды и кетоны Мұнай. Мұнайдың шығу тарихы
Мұнай. Мұнайдың шығу тарихы Строение атома. Периодический закон
Строение атома. Периодический закон Одноатомные и многоатомные спирты
Одноатомные и многоатомные спирты Окислительно-восстановительные реакции. 11 класс
Окислительно-восстановительные реакции. 11 класс Хімічні властивості кислот
Хімічні властивості кислот Чистые вещества и смеси. Способы разделения смесей
Чистые вещества и смеси. Способы разделения смесей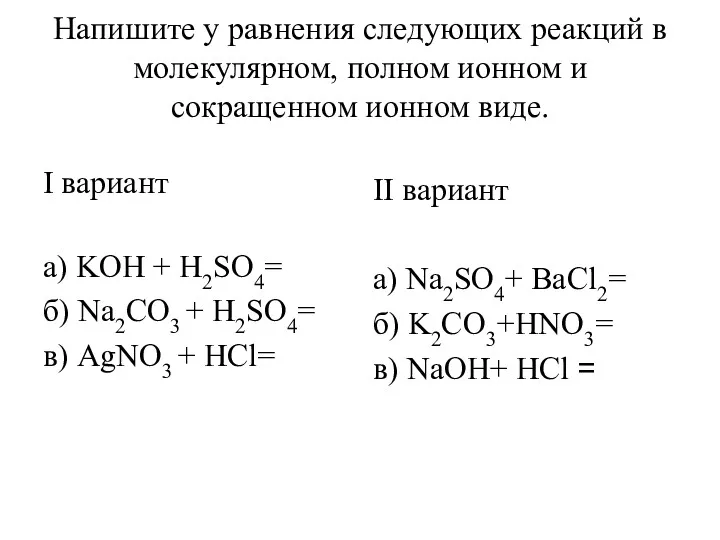 Уравнения реакций в молекулярном, полном ионном и сокращенном ионном виде
Уравнения реакций в молекулярном, полном ионном и сокращенном ионном виде Степень окисления
Степень окисления Закон действующих масс и его применение в аналитической химии
Закон действующих масс и его применение в аналитической химии Кислоты. Общая характеристика, химические свойства
Кислоты. Общая характеристика, химические свойства